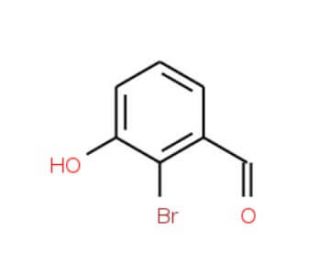
Molecular structure of 2-Bromo-3-hydroxybenzaldehyde, CAS Number: 196081-71-7
2-Bromo-3-hydroxybenzaldehyde (CAS 196081-71-7)
CAS Number:
196081-71-7
Molecular Weight:
201.02
Molecular Formula:
C7H5BrO2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-Bromo-3-hydroxybenzaldehyde is soluble in water and ethanol. This compound has found application in scientific research, notably in synthesizing novel compounds, probing biochemical pathways, and investigating chemical reactivity. As a reagent, it facilitates the synthesis of diverse compounds, including 2-bromo-3-hydroxybenzoic acid and 3-bromo-2-hydroxybenzoic acid. Additionally, it has been instrumental in studying biochemical pathways and exploring the reactivity of aldehydes with nucleophiles.
2-Bromo-3-hydroxybenzaldehyde (CAS 196081-71-7) References
- New chiral benzothiazine ligand and its use in the synthesis of a chiral receptor. | Harmata, M., et al. 2006. J Org Chem. 71: 3650-2. PMID: 16626160
- Enantioselective Synthesis of Both Epimers at C-21 in the Proposed Structure of Cytotoxic Macrolide Callyspongiolide. | Ghosh, AK. and Kassekert, LA. 2016. Org Lett. 18: 3274-7. PMID: 27331421
- Enantioselective total synthesis and structural assignment of callyspongiolide. | Ghosh, AK., et al. 2016. Org Biomol Chem. 14: 11357-11370. PMID: 27762414
- Enantioselective Synthesis of the Unsaturated Fragment of Callyspongiolide. | Matoušová, E., et al. 2016. Org Lett. 18: 5656-5659. PMID: 27783529
- Exploration of Biaryl Carboxylic Acids as Proton Shuttles for the Selective Functionalization of Indole C-H Bonds. | Pi, JJ., et al. 2018. J Org Chem. 83: 5791-5800. PMID: 29664291
- Structure Guided Design, Synthesis, and Biological Evaluation of Novel Benzosuberene Analogues as Inhibitors of Tubulin Polymerization. | Niu, H., et al. 2019. J Med Chem. 62: 5594-5615. PMID: 31059248
- Silver-Mediated Synthesis of Substituted Benzofuran- and Indole-Pyrroles via Sequential Reaction of ortho-Alkynylaromatics with Methylene Isocyanides. | Liu, JQ., et al. 2019. J Org Chem. 84: 8998-9006. PMID: 31117557
- Synthetic Studies toward Bazzanin K: Regioselective and Chemoselective Three-Component Suzuki Coupling. | Ju, X., et al. 2019. J Org Chem. 84: 12246-12252. PMID: 31448911
- Synthesis of the Tetracyclic Framework of Polycyclic Spiro Lignan Natural Products. | Ali, G. and Cuny, GD. 2020. ACS Omega. 5: 9007-9012. PMID: 32337465
- Synthesis and Structure-Activity Relationships of Aristoyagonine Derivatives as Brd4 Bromodomain Inhibitors with X-ray Co-Crystal Research. | Yoo, M., et al. 2021. Molecules. 26: PMID: 33802888
- Expanding Benzoxazole-Based Inosine 5'-Monophosphate Dehydrogenase (IMPDH) Inhibitor Structure-Activity As Potential Antituberculosis Agents. | Chacko, S., et al. 2021. J Med Chem. 64: 18233-18234. PMID: 34855408
- Alkyne Insertion Enabled Vinyl to Acyl 1,5-Palladium Migration: Rapid Access to Substituted 5-Membered-Dihydrobenzofurans and Indolines. | Ding, M., et al. 2023. Angew Chem Int Ed Engl. 62: e202300703. PMID: 36808789
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Bromo-3-hydroxybenzaldehyde, 1 g | sc-265449 | 1 g | $101.00 | |||
2-Bromo-3-hydroxybenzaldehyde, 5 g | sc-265449A | 5 g | $392.00 |
