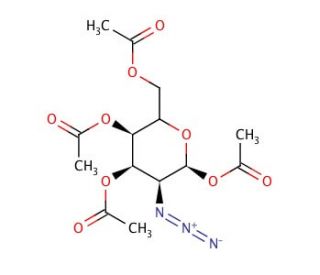
2-Azido-2-deoxy-1,3,4,6-tetra-O-acetyl-D-galactopyranose (CAS 84278-00-2)
QUICK LINKS
2-Azido-2-deoxy-1,3,4,6-tetra-O-acetyl-D-galactopyranose is a chemical compound widely employed in glycobiology research due to its significance in the synthesis of complex carbohydrates and glycoconjugates. Its acetylated form provides protection to the hydroxyl groups, facilitating selective chemical transformations at specific positions. This compound serves as a crucial building block for the preparation of glycosides, glycoproteins, and glycolipids, enabling researchers to investigate diverse biological processes involving carbohydrates. In research, 2-Azido-2-deoxy-1,3,4,6-tetra-O-acetyl-D-galactopyranose has been utilized for the synthesis of glycan probes for studying carbohydrate-protein interactions, such as lectin-carbohydrate binding assays and glycan microarray analysis. Additionally, its azido functional group allows for further derivatization via click chemistry, enabling the conjugation of various biomolecules, fluorophores, or nanoparticles to study cell surface glycans, intracellular trafficking, and glycan-mediated signaling pathways. Moreover, this compound has been instrumental in the development of glycosylation methodologies and the synthesis of glycomimetics for probing carbohydrate-mediated biological processes, including viral infection, cell adhesion, and immune response modulation. Overall, 2-Azido-2-deoxy-1,3,4,6-tetra-O-acetyl-D-galactopyranose plays a pivotal role in advancing our understanding of glycan biology and its implications in health and disease.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Azido-2-deoxy-1,3,4,6-tetra-O-acetyl-D-galactopyranose, 10 mg | sc-220704 | 10 mg | $330.00 |
