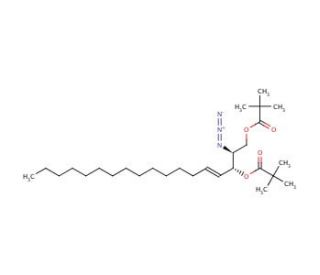
2-Azido-1,3-di-o-pivaloyl-D-erythro-sphingosine
QUICK LINKS
2-Azido-1,3-di-O-pivaloyl-D-erythro-sphingosine is a chemically modified sphingosine derivative, notable for its incorporation of azido groups and pivaloyl protections. This configuration makes it an exceptionally useful tool in biochemical research, particularly in the study of sphingolipid metabolism and the functional roles of sphingosine in cellular processes. The azido group introduces a reactive nitrogen functionality that can participate in click chemistry reactions, a method widely used to attach various probes or biomolecules in a highly specific and efficient manner. This feature allows for the precise labeling of the molecule in complex biological systems, enabling researchers to track its location and interactions within cells using various imaging techniques. The pivaloyl groups serve to protect the hydroxyl functions during synthetic modifications and can be removed under mild conditions to restore the natural hydroxyl groups, facilitating further biological activity. This compound has been used extensively to explore how sphingosine interacts with other cellular components, influencing signaling pathways and membrane dynamics. Investigations using 2-Azido-1,3-di-O-pivaloyl-D-erythro-sphingosine have provided valuable insights into the roles of sphingolipids in cell regulation, apoptosis, and the formation of lipid microdomains, contributing significantly to our understanding of lipid biochemistry and cell biology.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Azido-1,3-di-o-pivaloyl-D-erythro-sphingosine, 20 mg | sc-213763 | 20 mg | $330.00 |
