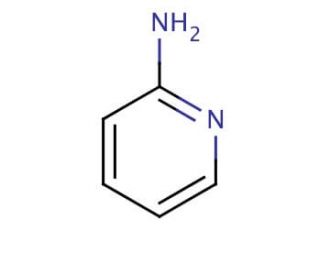
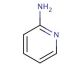
2-Aminopyridine (CAS 504-29-0)
See product citations (1)
QUICK LINKS
2-Aminopyridine functions as a versatile building block in organic synthesis. It acts as a nucleophilic reagent in various reactions, including nucleophilic aromatic substitution, metal-catalyzed cross-coupling reactions, and the synthesis of heterocyclic compounds. Its mechanism of action involves the activation of the amino group, allowing it to participate in nucleophilic attack on electrophilic substrates. 2-Aminopyridine also serves as a precursor for the synthesis of agrochemicals, and other fine chemicals. 2-Aminopyridine can undergo functionalization to introduce different substituents, making it useful for the preparation of diverse chemical compounds. Its role in organic synthesis is significant, as it enables the construction of complex molecular structures with specific functionalities.
2-Aminopyridine (CAS 504-29-0) References
- The benzoylation of 2-aminopyridine. | HUNTRESS, EH. and WALTER, HC. 1948. J Org Chem. 13: 735-7. PMID: 18884420
- 2-Aminopyridine derivatives as potential σ(2) receptor antagonists. | Abate, C., et al. 2012. ChemMedChem. 7: 1847-57. PMID: 22890883
- One-pot synthesis of 2-phenylimidazo[1,2-α]pyridines from acetophenone, [Bmim]Br(3) and 2-aminopyridine under solvent-free conditions. | Le, ZG., et al. 2012. Molecules. 17: 13368-75. PMID: 23143149
- Transition-metal-free access to 2-aminopyridine derivatives from 2-fluoropyridine and acetamidine hydrochloride. | Li, Y., et al. 2018. Org Biomol Chem. 16: 7564-7567. PMID: 30288539
- 2-Aminopyridine as a Nucleobase Substitute for Adenine in DNA-like Architectures: Synthesis of Alkynyl C-Nucleotides and Their Hybridization Characteristics. | Kurosaki, F., et al. 2020. J Org Chem. 85: 2666-2671. PMID: 31875396
- Discovery and optimization of 2-aminopyridine derivatives as novel and selective JAK2 inhibitors. | Ma, X., et al. 2020. Bioorg Med Chem Lett. 30: 127048. PMID: 32122740
- 2-Aminopyridine Analogs Inhibit Both Enzymes of the Glyoxylate Shunt in Pseudomonas aeruginosa. | McVey, AC., et al. 2020. Int J Mol Sci. 21: PMID: 32260167
- The 2-Aminopyridine Nucleobase Improves Triple-Helical Recognition of RNA and DNA When Used Instead of Pseudoisocytosine in Peptide Nucleic Acids. | Ryan, CA., et al. 2021. Biochemistry. 60: 1919-1925. PMID: 34097400
- 2-Aminopyridine - an unsung hero in drug discovery. | Rao, RN. and Chanda, K. 2022. Chem Commun (Camb). 58: 343-382. PMID: 34904599
- Cellular uptake of 2-aminopyridine-modified peptide nucleic acids conjugated with cell-penetrating peptides. | Brodyagin, N., et al. 2022. Biopolymers. 113: e23484. PMID: 34914092
- Efficient Synthesis of 2-Aminopyridine Derivatives: Antibacterial Activity Assessment and Molecular Docking Studies. | Kibou, Z., et al. 2022. Molecules. 27: PMID: 35684377
- Design, synthesis and biological evaluation of 2-aminopyridine derivatives as USP7 inhibitors. | Xu, X., et al. 2022. Bioorg Chem. 129: 106128. PMID: 36113266
- Investigating the X-aminopyridine (X = 2 and 3) molecules sensing by Al12N12 and B12N12 fullerene-like nanocages: DFT, QTAIM, RDG and TD-DFT insights. | Tamafo Fouégué, AD., et al. 2022. J Biomol Struct Dyn. 1-11. PMID: 36379673
- Room temperature phosphorescence of 2-aminopyridine with direct triplet state excitation. | Chavez, J., et al. 2023. Spectrochim Acta A Mol Biomol Spectrosc. 295: 122640. PMID: 36958243
- Excitation wavelength dependent S1-state decay dynamics of 2-aminopyridine and 3-aminopyridine. | Feng, B., et al. 2023. Phys Chem Chem Phys. 25: 17403-17409. PMID: 37350183
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Aminopyridine, 5 g | sc-213758 | 5 g | $20.00 | |||
2-Aminopyridine, 100 g | sc-213758A | 100 g | $41.00 |
