
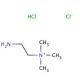
(2-Aminoethyl)trimethylammonium chloride hydrochloride (CAS 3399-67-5)
See product citations (2)
QUICK LINKS
(2-Aminoethyl)trimethylammonium chloride hydrochloride, known as ATMC-HCl, serves as an ionic surfactant in polymer synthesis and nanomaterial fabrication. It also acts as a reagent in biochemical and physiological studies. (2-Aminoethyl)trimethylammonium chloride hydrochloride assists in polymer synthesis by stabilizing the polymerization process and reducing reaction mixture viscosity. In nanomaterial fabrication, it enhances the dispersion of nanomaterials in aqueous solutions. Moreover, (2-Aminoethyl)trimethylammonium chloride hydrochloride serves as a catalyst in protein and other biomolecule reactions during biochemical and physiological studies. (2-Aminoethyl)trimethylammonium chloride hydrochloride, as a quaternary ammonium salt, showcases a trimethylammonium cation and a chloride anion. The cation has a large hydrophobic surface area, making it an effective surfactant. Conversely, the anion′s strong affinity for water molecules makes it an excellent stabilizer. The cation and anion interact to form an ionic bond, which is responsible for the surfactant and stabilizing properties of (2-Aminoethyl)trimethylammonium chloride hydrochloride.
(2-Aminoethyl)trimethylammonium chloride hydrochloride (CAS 3399-67-5) References
- Coating of adenovirus type 5 with polymers containing quaternary amines prevents binding to blood components. | Subr, V., et al. 2009. J Control Release. 135: 152-8. PMID: 19166885
- Room temperature, aqueous post-polymerization modification of glycidyl methacrylate-containing polymer brushes prepared via surface-initiated atom transfer radical polymerization. | Barbey, R. and Klok, HA. 2010. Langmuir. 26: 18219-30. PMID: 21062007
- Dicationic ion-pairing of phosphoric acid diesters post-liquid chromatography and subsequent determination by electrospray positive ionization-tandem mass spectrometry. | Chu, S., et al. 2011. J Chromatogr A. 1218: 8083-8. PMID: 21945623
- Cationic labeling of oligosaccharides for electrophoretic preconcentration and separation with contactless conductivity detection. | Partyka, J. and Foret, F. 2012. J Chromatogr A. 1267: 116-20. PMID: 22770384
- Design of multifunctional gold nanoparticles for in vitro and in vivo gene silencing. | Conde, J., et al. 2012. ACS Nano. 6: 8316-24. PMID: 22882598
- Enhanced electron transfer dissociation of peptides modified at C-terminus with fixed charges. | Ko, BJ. and Brodbelt, JS. 2012. J Am Soc Mass Spectrom. 23: 1991-2000. PMID: 22895859
- Charge-controlled nanoprecipitation as a modular approach to ultrasmall polymer nanocarriers: making bright and stable nanoparticles. | Reisch, A., et al. 2015. ACS Nano. 9: 5104-16. PMID: 25894117
- Basic Physicochemical Properties of Polyethylene Glycol Coated Gold Nanoparticles that Determine Their Interaction with Cells. | Del Pino, P., et al. 2016. Angew Chem Int Ed Engl. 55: 5483-7. PMID: 27028669
- Derivatization enhanced separation and sensitivity of long chain-free fatty acids: Application to asthma using targeted and non-targeted liquid chromatography-mass spectrometry approach. | Bian, X., et al. 2017. Anal Chim Acta. 989: 59-70. PMID: 28915943
- Formation of pH-Resistant Monodispersed Polymer-Lipid Nanodiscs. | Ravula, T., et al. 2018. Angew Chem Int Ed Engl. 57: 1342-1345. PMID: 29232017
- Investigation of a side reaction occurring during N-linked glycan labeling by cationic tags. | Krenkova, J., et al. 2018. J Chromatogr A. 1570: 67-74. PMID: 30086833
- Isothiocyanate-Functionalized Mesoporous Silica Nanoparticles as Building Blocks for the Design of Nanovehicles with Optimized Drug Release Profile. | Martínez-Edo, G., et al. 2019. Nanomaterials (Basel). 9: PMID: 31470557
- No-additive salting-out liquid-liquid extraction-A tool for purification of positively charged compounds from highly salted reaction mixtures. | Moravcová, D., et al. 2020. J Sep Sci. 43: 4356-4363. PMID: 33021338
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(2-Aminoethyl)trimethylammonium chloride hydrochloride, 1 g | sc-237910 | 1 g | $215.00 | |||
(2-Aminoethyl)trimethylammonium chloride hydrochloride, 5 g | sc-237910A | 5 g | $921.00 |
