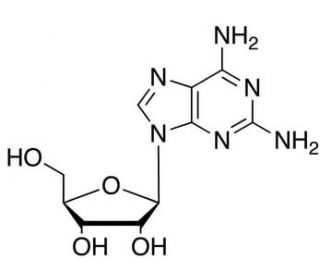
2-Amino Adenosine (CAS 2096-10-8)
See product citations (1)
QUICK LINKS
2-Amino Adenosine is a purine nucleoside that functions as a precursor in the biosynthesis of adenosine, a crucial component of RNA and ATP. It plays a role in the regulation of cellular metabolism and energy production by participating in the synthesis of adenosine, which is essential for various cellular processes. At the molecular level, 2-Amino Adenosine interacts with enzymes involved in the adenosine biosynthetic pathway, influencing the production of adenosine and its subsequent utilization in cellular functions. This chemical compound modulates the activity of adenosine kinase, an enzyme responsible for the phosphorylation of adenosine to form AMP, thereby impacting the intracellular levels of adenosine and its derivatives. Through its involvement in the adenosine metabolic pathway, 2-Amino Adenosine contributes to the regulation of cellular energy metabolism and nucleic acid synthesis, playing a specific role in the molecular processes underlying these cellular functions.
2-Amino Adenosine (CAS 2096-10-8) References
- 7-Functionalized 7-deazapurine ribonucleosides related to 2-aminoadenosine, guanosine, and xanthosine: glycosylation of pyrrolo[2,3-d]pyrimidines with 1-O-acetyl-2,3,5-tri-O-benzoyl-D-ribofuranose. | Seela, F. and Peng, X. 2006. J Org Chem. 71: 81-90. PMID: 16388621
- Antisense probes containing 2-aminoadenosine allow efficient depletion of U5 snRNP from HeLa splicing extracts. | Lamm, GM., et al. 1991. Nucleic Acids Res. 19: 3193-8. PMID: 1648201
- An efficient process for synthesis of 2'-O-methyl and 3'-O-methyl guanosine from 2-aminoadenosine using diazomethane and the catalyst stannous chloride. | Kore, AR., et al. 2006. Nucleosides Nucleotides Nucleic Acids. 25: 307-14. PMID: 16629123
- New synthetic routes to synthons suitable for 2'-O-allyloligoribonucleotide assembly. | Sproat, BS., et al. 1991. Nucleic Acids Res. 19: 733-8. PMID: 1708121
- Pyrazolo[3,4-d]pyrimidine ribonucleosides related to 2-aminoadenosine and isoguanosine: synthesis, deamination and tautomerism. | Seela, F. and Xu, K. 2007. Org Biomol Chem. 5: 3034-45. PMID: 17728871
- Synthesis study of 2'-O-(2-methoxyethyl)-purine derivatives. | Sivets, GG. 2007. Nucleosides Nucleotides Nucleic Acids. 26: 1237-40. PMID: 18066759
- One-electron reduction of 8-bromo-2-aminoadenosine in the aqueous phase: radiation chemical and DFT studies of the mechanism. | Kaloudis, P., et al. 2008. J Phys Chem B. 112: 5209-17. PMID: 18373377
- Use of base-modified duplex-stabilizing deoxynucleoside 5'-triphosphates to enhance the hybridization properties of primers and probes in detection polymerase chain reaction. | Kutyavin, IV. 2008. Biochemistry. 47: 13666-73. PMID: 19046073
- [Affinity capture of specific DNA fragments with the use of short synthetic sequences]. | Mikhaĭlov, VS., et al. 2013. Bioorg Khim. 39: 81-6. PMID: 23844509
- Expanding a fluorescent RNA alphabet: synthesis, photophysics and utility of isothiazole-derived purine nucleoside surrogates. | Rovira, AR., et al. 2017. Chem Sci. 8: 2983-2993. PMID: 28451365
- Dye-conjugated complementary lipophilic nucleosides as useful probes to study association processes by fluorescence resonance energy transfer. | Mayoral, MJ., et al. 2017. Org Biomol Chem. 15: 7558-7565. PMID: 28857106
- Fluorescing Isofunctional Ribonucleosides: Assessing Adenosine Deaminase Activity and Inhibition. | Ludford, PT., et al. 2019. Chembiochem. 20: 718-726. PMID: 30566279
- Intrinsic Contributions of 2'-Hydroxyl to the Hydration of Nucleosides at the Monomeric Level. | Wang, Z., et al. 2020. Chemistry. 26: 17046-17055. PMID: 32786015
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Amino Adenosine, 25 mg | sc-220693 | 25 mg | $62.00 | |||
2-Amino Adenosine, 200 mg | sc-220693A | 200 mg | $124.00 |
