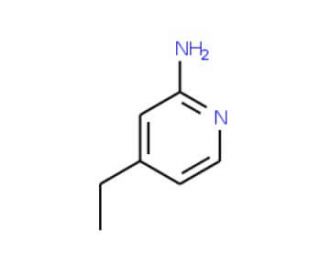
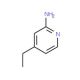
2-Amino-4-ethylpyridine (CAS 33252-32-3)
QUICK LINKS
2-Amino-4-ethylpyridine, registered under the CAS number 33252-32-3, is a chemical compound belonging to the class of organic compounds known as pyridines. This compound features a pyridine ring—a heterocyclic ring consisting of five carbon atoms and one nitrogen atom—substituted by an amino group at the 2-position and an ethyl group at the 4-position. The presence of both amino and ethyl groups on the pyridine ring enhances its chemical reactivity and confers unique properties that make it valuable in various research applications, particularly in organic synthesis and catalysis. In research settings, 2-Amino-4-ethylpyridine is often utilized as a building block for the synthesis of more complex molecules. Its amino group can act as a nucleophile, enabling the formation of bonds with electrophilic species, which is a fundamental reaction in constructing nitrogen-containing heterocycles that are prevalent in many biologically active compounds. Additionally, the basic nature of the amino group allows it to function as a ligand in coordination chemistry, forming complexes with metals, which can be used in catalysis or materials science. Studies involving this compound help in understanding the interactions and transformations of pyridine derivatives in chemical reactions, providing insights that are essential for advancing synthetic methodologies and developing new materials with specific desired properties.
2-Amino-4-ethylpyridine (CAS 33252-32-3) References
- Design and synthesis of orally bioavailable inhibitors of inducible nitric oxide synthase. Part 1: synthesis and biological evaluation of dihydropyridin-2-imines. | Kawanaka, Y., et al. 2002. Bioorg Med Chem Lett. 12: 2291-4. PMID: 12161118
- Additional steps toward molecular scale wires: Further study of Ni(5)(10/11+) chains embraced by polypyridylamide ligands. | Berry, JF., et al. 2003. Inorg Chem. 42: 3534-9. PMID: 12767190
- Enhancing the stability of trinickel molecular wires and switches: Ni(3)(6+)/Ni(3)(7+). | Berry, JF., et al. 2003. Inorg Chem. 42: 3595-601. PMID: 12767198
- Molecular recognition of carbohydrates with acyclic pyridine-based receptors. | Mazik, M., et al. 2004. J Org Chem. 69: 7448-62. PMID: 15497969
- Discovery and analgesic evaluation of 8-chloro-1,4-dihydropyrido[2,3-b]pyrazine-2,3-dione as a novel potent d-amino acid oxidase inhibitor. | Xie, D., et al. 2016. Eur J Med Chem. 117: 19-32. PMID: 27089209
- The preparation of some substituted 2, 6-bis-(2-pyridyl)-pyridines. | Case, et al. 1956. Journal of the American Chemical Society. 78.22: 5842-5844.
- 1, 3-Shifts. V. The Intermolecular Nature of Some 1, 3-Shifts. | Wiberg, et al. 1957. Journal of the American Chemical Society. 79.12: 3160-3164.
- Catalytic Synthesis of Heterocycles. 1 X. Dehydrocyclization of 4-Ethyl-3-pyridinethiol to 6-Azathianaphthene. | HANSCH, et al. 1958. The Journal of Organic Chemistry. 23.12: 1924-1926.
- Preparation of substituted 2-oxazolidones from 1, 2-epoxides and isocyanates. | Speranza, et al. 1958. The Journal of Organic Chemistry. 23.12: 1922-1924.
- Charge-transfer complexes formed in the reaction of 2-amino-4-ethylpyridine with π-electron acceptors. | AlQaradawi, et al. 2016. Journal of Molecular Structure. 1106: 10-18.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Amino-4-ethylpyridine, 1 g | sc-209039 | 1 g | $380.00 |
