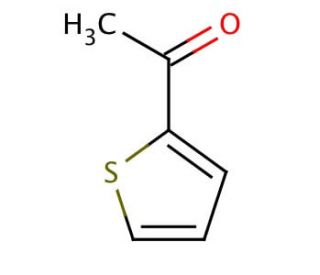
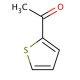
2-Acetylthiophene (CAS 88-15-3)
QUICK LINKS
2-Acetylthiophene (2-AT) stands as a heterocyclic compound, forming a five-membered ring and comprising two carbon atoms, two sulfur atoms, and an oxygen atom. Its significance lies in its role as a pivotal intermediate during the synthesis of a range of compounds, including dyes and fragrances. The versatility of 2-Acetylthiophene has prompted extensive examination of its potential applications within the realms of medicine and biochemistry. The exact mechanism by which 2-Acetylthiophene operates remains partially veiled. Nonetheless, it is posited that the compound binds to specific receptors within the body, thereby instigating a sequence of biochemical and physiological reactions. These orchestrated responses are likely accountable for the diverse impacts attributed to 2-Acetylthiophene.
2-Acetylthiophene (CAS 88-15-3) References
- Solvent effects on infrared spectra of 2-acetylthiophene in organic solvents. | Liu, Q., et al. 2003. Spectrochim Acta A Mol Biomol Spectrosc. 59: 471-5. PMID: 12524117
- Infrared study on solvent-solute interactions of 2-acetylthiophene in binary mixtures. | Liu, Q., et al. 2004. Spectrochim Acta A Mol Biomol Spectrosc. 60: 397-400. PMID: 14670504
- Rapid detection and characterization of minor reactive metabolites using stable-isotope trapping in combination with tandem mass spectrometry. | Yan, Z., et al. 2005. Rapid Commun Mass Spectrom. 19: 3322-30. PMID: 16235238
- Synthesis, characterization, electro chemistry, catalytic and biological activities of ruthenium(III) complexes with bidentate N, O/S donor ligands. | Balasubramanian, KP., et al. 2006. Spectrochim Acta A Mol Biomol Spectrosc. 65: 678-83. PMID: 16546440
- Generation of a structurally diverse library through alkylation and ring closure reactions using 3-dimethylamino-1-(thiophen-2-yl)propan-1-one hydrochloride. | Roman, G. 2013. Acta Chim Slov. 60: 70-80. PMID: 23841334
- Apoptotic effect of chalcone derivatives of 2-acetylthiophene in human breast cancer cells. | Fogaça, TB., et al. 2017. Pharmacol Rep. 69: 156-161. PMID: 27923159
- DFT study of the interactions between thiophene-based corrosion inhibitors and an Fe4 cluster. | Dao, DQ., et al. 2017. J Mol Model. 23: 260. PMID: 28801757
- Synthesis and evaluation of novel substituted 1,2,3-triazolyldihydroquinolines as promising antitubercular agents. | Marvadi, SK., et al. 2019. Bioorg Med Chem Lett. 29: 529-533. PMID: 30638877
- Domino Synthetic Strategy for Tetrahydrothiopyran Derivatives from Benzaldehydes, 2-Acetylfuran/2-Acetylthiophene, and Sodium Sulfide. | Chen, D., et al. 2020. J Org Chem. 85: 9088-9095. PMID: 32530280
- Condensed-phase relative Gibbs free energy and E/Z descriptors for 2-acetylthiophene and 2-acetylthiophene-N1-phenyl thiosemicarbazones. | Sales, ALR., et al. 2021. J Mol Model. 27: 101. PMID: 33660108
- Determination of the semiexperimental equilibrium structure of 2-acetylthiophene in the presence of methyl internal rotation and substituent effects compared to thiophene. | Dindić, C., et al. 2022. Phys Chem Chem Phys. 24: 3804-3815. PMID: 35084004
- RIFM fragrance ingredient safety assessment, 2-acetylthiophene, CAS registry number 88-15-3. | Api, AM., et al. 2023. Food Chem Toxicol. 176 Suppl 1: 113704. PMID: 36889427
- Cinchona-Alkaloid-Derived NN Ligands and Achiral Phosphines for Iridium-Catalyzed Asymmetric Hydrogenation of Heteroaromatic and α-Chloroheteroaryl Ketones. | Tian, J., et al. 2023. J Org Chem. 88: 9213-9224. PMID: 37226800
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Acetylthiophene, 25 g | sc-237897 | 25 g | $37.00 |
