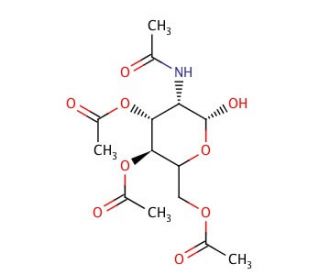
2-(Acetylamino)-2-deoxy-D-glucopyranose 3,4,6-Triacetate (CAS 34051-43-9)
QUICK LINKS
2-(Acetylamino)-2-deoxy-D-glucopyranose 3,4,6-triacetate, a derivative of D-glucopyranose, finds extensive utility in glycobiology research owing to its role in the synthesis and manipulation of complex carbohydrates. This compound serves as a key building block for generating diverse glycan structures and glycoconjugates. Its triacetate form facilitates chemical modifications by protecting hydroxyl groups, enabling precise control over glycan synthesis and functionalization. Researchers utilize this chemical in the development of glycan microarrays, where it serves as a probe for studying glycan-protein interactions and cell surface recognition events. Additionally, it plays a crucial role in elucidating glycan biosynthesis pathways and glycosylation processes by serving as a substrate for glycosyltransferase enzymes involved in glycan assembly. Furthermore, this compound is employed in structural characterization studies using techniques like NMR spectroscopy and mass spectrometry, aiding in the identification and analysis of complex glycan structures. Overall, 2-(acetylamino)-2-deoxy-D-glucopyranose 3,4,6-triacetate serves as a versatile tool in glycobiology research, enabling the investigation of glycan-mediated biological processes and facilitating the development of novel glycan-based technologies.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-(Acetylamino)-2-deoxy-D-glucopyranose 3,4,6-Triacetate, 25 mg | sc-208932 | 25 mg | $300.00 |
