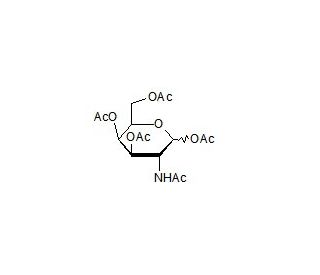
2-Acetamido-2-deoxy-D-galactopyranose-1,3,4,6-tetra-O-acetate (CAS 76375-60-5)
QUICK LINKS
2-Acetamido-2-deoxy-D-galactopyranose-1,3,4,6-tetra-O-acetate is a derivative of D-galactopyranose, commonly utilized in carbohydrate chemistry and glycobiology research. This compound serves as a building block for synthesizing complex oligosaccharides and glycoconjugates, particularly those containing galactose residues. Due to its versatile chemical structure, researchers employ it in the synthesis of oligosaccharide libraries for investigating carbohydrate-protein interactions, such as lectin binding studies and glycan microarray analysis. Additionally, 2-Acetamido-2-deoxy-D-galactopyranose-1,3,4,6-tetra-O-acetate finds application in the development of glycosylation methodologies and glycosyltransferase enzyme assays, facilitating the study of glycosylation pathways and glycan biosynthesis. Moreover, this compound plays a crucial role in the synthesis of glycolipids and glycosaminoglycans, contributing to our understanding of their structure-function relationships and biological roles. By providing researchers with a synthetic tool to access complex glycan structures, 2-Acetamido-2-deoxy-D-galactopyranose-1,3,4,6-tetra-O-acetate advances our knowledge of carbohydrate biology and aids in the development of glycan-based diagnostics.
2-Acetamido-2-deoxy-D-galactopyranose-1,3,4,6-tetra-O-acetate (CAS 76375-60-5) References
- Altered glycosylation of membrane glycoproteins associated with human mammary carcinoma. | Hiraizumi, S., et al. 1992. Jpn J Cancer Res. 83: 1063-72. PMID: 1452459
- A highly alpha-selective glycosylation for the convenient synthesis of repeating alpha-(1-->4)-linked N-acetyl-galactosamine units. | Yang, L. and Ye, XS. 2010. Carbohydr Res. 345: 1713-21. PMID: 20591420
- Targeting hepatic cancer cells with pegylated dendrimers displaying N-acetylgalactosamine and SP94 peptide ligands. | Medina, SH., et al. 2013. Adv Healthc Mater. 2: 1337-50. PMID: 23554387
- Chemoenzymatic synthesis of sulfur-linked sugar polymers as heparanase inhibitors. | He, P., et al. 2022. Nat Commun. 13: 7438. PMID: 36460670
- Curcuma wenyujin rhizomes extract ameliorates lipid accumulation. | Wang, H., et al. 2024. Fitoterapia. 175: 105957. PMID: 38604260
- Appraisal of a glycopeptide cloaking strategy for a therapeutic oligopeptide: glycopeptide analogs of the renin inhibitor ditekiren. | Harrison, AW., et al. 1994. Bioorg Med Chem. 2: 1339-61. PMID: 7788297
- Acetohalogeno Derivatives and Glycosides of D-Galactosamine1. | Tarasiejska, Zofia and Roger W. Jeanloz. 1958. Journal of the American Chemical Society. 80.23: 6325-6327.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Acetamido-2-deoxy-D-galactopyranose-1,3,4,6-tetra-O-acetate, 250 mg | sc-220681 | 250 mg | $202.00 |
