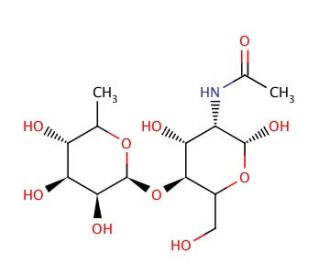
2-Acetamido-2-deoxy-4-O-(α-L-fucopyranosyl)-D-glucopyranose (CAS 76211-71-7)
QUICK LINKS
2-Acetamido-2-deoxy-4-O-(α-L-fucopyranosyl)-D-glucopyranose, also known as Fuc-α-2-4-GlcNAc, is a glycoside compound commonly used in glycobiology research. It consists of a D-glucopyranose moiety linked to an α-L-fucopyranosyl residue via an O-glycosidic bond, with an acetamido group attached to the C2 position of the glucosamine unit. This chemical mimics the structural motifs found in complex glycans and glycoconjugates present on cell surfaces and extracellular matrices. Researchers utilize Fuc-α-2-4-GlcNAc as a molecular probe to investigate carbohydrate-binding interactions involved in various biological processes, including cell adhesion, signaling, and immune recognition. Its application extends to studies of lectins, antibodies, and carbohydrate-processing enzymes, such as glycosyltransferases and glycosidases. Furthermore, Fuc-α-2-4-GlcNAc serves as a valuable tool in glycomics and glycoproteomics research, facilitating the characterization and quantification of glycan structures in cells, tissues, and biological fluids. By elucidating the roles of specific glycans in health and disease, Fuc-α-2-4-GlcNAc contributes to advancing our understanding of glycan-mediated cellular functions and offers insights into targeting carbohydrate-based interactions.
2-Acetamido-2-deoxy-4-O-(α-L-fucopyranosyl)-D-glucopyranose (CAS 76211-71-7) References
- Structural basis for the interaction between human milk oligosaccharides and the bacterial lectin PA-IIL of Pseudomonas aeruginosa. | Perret, S., et al. 2005. Biochem J. 389: 325-32. PMID: 15790314
- X-ray structures and thermodynamics of the interaction of PA-IIL from Pseudomonas aeruginosa with disaccharide derivatives. | Marotte, K., et al. 2007. ChemMedChem. 2: 1328-38. PMID: 17623286
- The first structure-function study of GH151 α-l-fucosidase uncovers new oligomerization pattern, active site complementation, and selective substrate specificity. | Koval'ová, T., et al. 2022. FEBS J. 289: 4998-5020. PMID: 35113503
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Acetamido-2-deoxy-4-O-(α-L-fucopyranosyl)-D-glucopyranose, 5 mg | sc-220677 | 5 mg | $330.00 |
