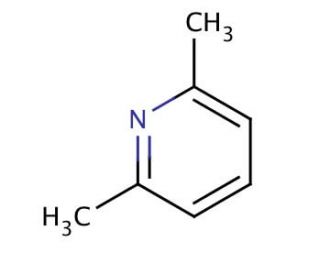
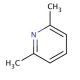
Molecular structure of 2,6-Lutidine, CAS Number: 108-48-5
2,6-Lutidine (CAS 108-48-5)
CAS Number:
108-48-5
Molecular Weight:
107.15
Molecular Formula:
C7H9N
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,6-Lutidine is an aromatic heterocyclic compound, demonstrating solubility in various organic solvents. This compound plays a role as an intermediate in the production of diverse dyes and other chemical substances. Moreover, it holds broad application in synthesizing other aromatic compounds, including 2,6-dihydropyridine and 2,6-dimethoxy-pyridine.
2,6-Lutidine (CAS 108-48-5) References
- Kilo-scale synthesis process for 2'-O-(2-methoxyethyl)-pyrimidine derivatives. | Ross, BS., et al. 2005. Nucleosides Nucleotides Nucleic Acids. 24: 815-8. PMID: 16248042
- A series of mononuclear quasi-two-coordinate copper(I) complexes employing a sterically demanding thiolate ligand. | Groysman, S. and Holm, RH. 2009. Inorg Chem. 48: 621-7. PMID: 19138143
- Micelles of a diblock copolymer of styrene and ethylene oxide in mixtures of 2,6-lutidine and water. | Tuzar, Z., et al. 2008. Langmuir. 24: 13863-5. PMID: 19360951
- Copper-catalyzed, aerobic oxidative cross-coupling of alkynes with arylboronic acids: remarkable selectivity in 2,6-lutidine media. | Yasukawa, T., et al. 2011. Org Biomol Chem. 9: 6208-10. PMID: 21808793
- Oxoammonium salt oxidations of alcohols in the presence of pyridine bases. | Bobbitt, JM., et al. 2014. J Org Chem. 79: 1055-67. PMID: 24386938
- Electrophilic Phosphonium Cation-Mediated Phosphane Oxide Reduction Using Oxalyl Chloride and Hydrogen. | Stepen, AJ., et al. 2018. Angew Chem Int Ed Engl. 57: 15253-15256. PMID: 30230149
- Bis(pentalene)dititanium chemistry: C-H, C-X and H-H bond activation. | Tsoureas, N., et al. 2018. Dalton Trans. 47: 14531-14539. PMID: 30252008
- Access to Stereodefined (E)-2-Silylallylboronates via Regioselective Chloroboration of Allenylsilanes. | Yang, Z., et al. 2019. Org Lett. 21: 9541-9544. PMID: 31713429
- Photochemical Conversion of Isoxazoles to 5-Hydroxyimidazolines. | Morita, T., et al. 2020. Org Lett. 22: 3460-3463. PMID: 32286839
- Mechanistic details of metal-free cyclization reaction of organophosphorus oxide with alkynes mediated by 2,6-lutidine and Tf2 O. | Tian, J., et al. 2020. J Comput Chem. 41: 1709-1717. PMID: 32323872
- Confined Water Molecules in Binary Mixtures of Water and 2,6-Lutidine Near Lower Solution Critical Temperature. | Korotkevich, AA. and Bakker, HJ. 2021. J Phys Chem B. 125: 287-296. PMID: 33370126
- Calcium-mediated C(sp3)-H Activation and Alkylation of Alkylpyridines. | Zheng, X., et al. 2021. Inorg Chem. 60: 5114-5121. PMID: 33728911
- Photoredox-catalyzed intramolecular cyclopropanation of alkenes with α-bromo-β-keto esters. | Ide, K., et al. 2021. Org Biomol Chem. 19: 9172-9176. PMID: 34664610
- Efficient Purification of 2,6-Lutidine by Nonporous Adaptive Crystals of Pillararenes. | Wang, Z., et al. 2022. ACS Appl Mater Interfaces. 14: 41072-41078. PMID: 36053117
- Synthesis of β,β-Disubstituted Styrenes via Trimethylsilyl Trifluoromethanesulfonate-Promoted Aldehyde-Aldehyde Aldol Coupling-Elimination. | Dixon, GJ., et al. 2022. J Org Chem. 87: 14846-14854. PMID: 36239694
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,6-Lutidine, 25 ml | sc-238428 | 25 ml | $89.00 |
