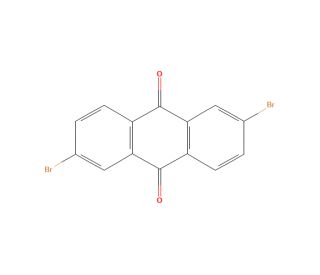
2,6-Dibromoanthraquinone (CAS 633-70-5)
QUICK LINKS
2,6-Dibromoanthraquinone (2,6-DBQ) is an organic compound belonging to the family of anthraquinones. It is a useful intermediate in the synthesis of a variety of organic compounds, such as dyes and pigments. It can also be used as a probe or marker in chemical analysis, chromatography, or fluorescence-based studies. Furthermore, 2,6-Dibromoanthraquinone is applied as a component of organic light-emitting diodes (OLEDs). This compound is an electron acceptor, which enables it to initiate or catalyze a variety of reactions, such as the oxidation of organic compounds, the formation of polymers networks, and the synthesis of organic compounds. Studies have reported antioxidant, anti-inflammatory, anticancer, antibacterial, and antifungal properties of 2,6-Dibromoanthraquinone.
2,6-Dibromoanthraquinone (CAS 633-70-5) References
- Synthesis and properties of an anthraquinone-based redox switch for molecular electronics. | van Dijk, EH., et al. 2006. Org Lett. 8: 2333-6. PMID: 16706519
- Long-wavelength analogue of PRODAN: synthesis and properties of Anthradan, a fluorophore with a 2,6-donor-acceptor anthracene structure. | Lu, Z., et al. 2006. J Org Chem. 71: 9651-7. PMID: 17168582
- Soluble and easily crystallized anthracene derivatives: precursors of solution-processable semiconducting molecules. | Park, JH., et al. 2007. Org Lett. 9: 2573-6. PMID: 17542599
- Synthesis and electrochemical evaluation of conjugates between 2'-deoxyadenosine and modified anthraquinone: probes for hole-transfer studies in DNA. | Abou-Elkhair, RA., et al. 2009. J Org Chem. 74: 4712-9. PMID: 19507820
- High efficient organic light emitting diodes using new 9, 10-diphenylanthracene derivatives containing bulky substituents on 2, 6-positon | Jo, W. J., Kim, K. H., No, H. C., Shin, D. Y., Oh, S. J., Son, J. H.,.. & Kwon, S. K. 2009. Synthetic metals. 159(13): 1359-1364.
- Synthesis and characterization of a new series of blue fluorescent 2, 6-linked 9, 10-diphenylanthrylenephenylene copolymers and their application for polymer light-emitting diodes | Chen, H. Y., Chen, C. T., & Chen, C. T. 2010. Macromolecules. 43(8): 3613-3623.
- Synthesis and characterisation of liquid crystalline anthraquinone dyes with excellent dichroism and solubility | Zhang, W., Zhang, C., Chen, K., Wang, Z., Wang, M., Ding, H.,.. & Yang, H. 2016. Liquid Crystals. 43(10): 1307-1314.
- Selective adsorption of a supramolecular structure on flat and stepped gold surfaces | Peköz, R., & Donadio, D. 2018. Surface Science. 670: 44-50.
- New anthraquinone‐based conjugated microporous polymer cathode with ultrahigh specific surface area for high‐performance lithium‐ion batteries | Molina, A., Patil, N., Ventosa, E., Liras, M., Palma, J., & Marcilla, R. 2020. Advanced Functional Materials. 30(6): 1908074.
- Electrochemical Study of Poly (2, 6‐Anthraquinonyl Sulfide) as Cathode for Alkali‐Metal‐Ion Batteries | Hu, Y., Gao, Y., Fan, L., Zhang, Y., Wang, B., Qin, Z.,.. & Lu, B. 2020. Advanced Energy Materials. 10(48): 2002780.
- Anthraquinone-enriched conjugated microporous polymers as organic cathode materials for high-performance lithium-ion batteries | Mohamed, M. G., Sharma, S. U., Yang, C. H., Samy, M. M., Mohammed, A. A., Chaganti, S. V.,.. & Wei-Kuo, S. 2021. ACS Applied Energy Materials. 4(12): 14628-14639.
- Conjugated Organic Polymers with Anthraquinone Redox Centers for Efficient Photocatalytic Hydrogen Peroxide Production from Water and Oxygen under Visible Light Irradiation without Any Additives | Xu, X., Sa, R., Huang, W., Sui, Y., Chen, W., Zhou, G.,.. & Zhong, H. 2022. ACS Catalysis. 12(20): 12954-12963.
- Poly (2, 6-anthraquinonyl disulfide) as a high-capacity and high-power cathode for rechargeable magnesium batteries: extra capacity provided by the disulfide group | Ren, X., Tao, D., Tang, Y., Cao, Y., & Xu, F. 2023. Journal of Materials Chemistry A. 11(18): 9955-9963.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,6-Dibromoanthraquinone, 1 g | sc-482150 | 1 g | $274.00 |
