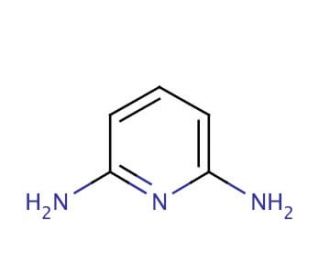
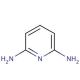
2,6-Diaminopyridine (CAS 141-86-6)
QUICK LINKS
2,6-Diaminopyridine functions as a reagent in chemical synthesis. It acts as a building block in the formation of various organic compounds, particularly in the production of agrochemicals, and specialty chemicals. The mechanism of action involves its ability to participate in nucleophilic substitution reactions, where it can undergo substitution with other functional groups to form new chemical bonds. Its role in chemical synthesis involves its interaction at the molecular level, where it can facilitate the formation of specific chemical structures through its reactivity with other compounds. 2,6-Diaminopyridine′s mechanism of action is characterized by its ability to undergo chemical transformations, leading to the production of diverse organic compounds with potential applications in various fields of research and development.
2,6-Diaminopyridine (CAS 141-86-6) References
- Derivatives of 2,6-diaminopyridine. | BERNSTEIN, J. and STEARNS, B. 1947. J Am Chem Soc. 69: 1151-8. PMID: 20240506
- Macrocyclic metal complexes derived from 2,6-diaminopyridine and isatin with their antibacterial and spectroscopic studies. | Singh, DP., et al. 2010. Spectrochim Acta A Mol Biomol Spectrosc. 76: 45-9. PMID: 20338803
- Synthesis, characterization and biological activities of some new chromium molybdenum and tungsten complexes with 2,6-diaminopyridine. | Soliman, AA., et al. 2012. Spectrochim Acta A Mol Biomol Spectrosc. 89: 329-32. PMID: 22286054
- The role of 2,6-diaminopyridine ligands in the isolation of an unprecedented, low-valent tin complex. | Flock, J., et al. 2013. Chemistry. 19: 15504-17. PMID: 24591247
- 2,6-Diaminopyridine-imprinted polymer and its potency to hair-dye assay using graphene/ionic liquid electrochemical sensor. | Zhao, P. and Hao, J. 2015. Biosens Bioelectron. 64: 277-84. PMID: 25441409
- High-spin iron(II) complexes with mono-phosphorylated 2,6-diaminopyridine ligands. | Schröder-Holzhacker, C., et al. 2016. Monatsh Chem. 147: 1539-1545. PMID: 27546911
- Synergistic effect of tartaric acid with 2,6-diaminopyridine on the corrosion inhibition of mild steel in 0.5 M HCl. | Qiang, Y., et al. 2016. Sci Rep. 6: 33305. PMID: 27628901
- Crystal structure of pharmaceutical cocrystals of 2,6-diaminopyridine with piracetam and theophylline. | Durán-Palma, MH., et al. 2017. Acta Crystallogr C Struct Chem. 73: 767-772. PMID: 28978781
- A new polymorph of 2,6-diaminopyridine. | Hall, VM., et al. 2017. Acta Crystallogr C Struct Chem. 73: 990-993. PMID: 29111531
- Polyamine-co-2, 6-diaminopyridine covalently bonded on chitosan for the adsorptive removal of Hg(II) ions from aqueous solution. | Liang, W., et al. 2019. Int J Biol Macromol. 130: 853-862. PMID: 30840868
- Excellent Toughening of 2,6-Diaminopyridine Derived Poly (Urethane Urea) via Dynamic Cross-Linkages and Interfering with Hydrogen Bonding of Urea Groups from Partially Coordinated Ligands. | Sun, A., et al. 2019. Polymers (Basel). 11: PMID: 31394877
- Synthesis, Structural Characterization, Enzymatic and Oxidative Polymerization of 2,6-Diaminopyridine. | Şenol, D. 2020. J Fluoresc. 30: 157-174. PMID: 31970584
- Effect of Substitution at Amine Functionality of 2,6-Diaminopyridine-Coupled Rhodamine on Metal-Ion Interaction and Self-Assembly. | Panja, S., et al. 2020. ACS Omega. 5: 13984-13993. PMID: 32566865
- 2,6-Diaminopyridine-Based Polyurea as an ORR Electrocatalyst of an Anion Exchange Membrane Fuel Cell. | Wang, YZ., et al. 2023. Polymers (Basel). 15: PMID: 36850199
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,6-Diaminopyridine, 25 g | sc-225761 | 25 g | $47.00 |
