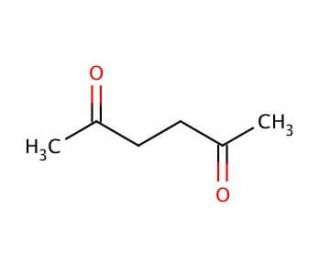
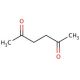
Molecular structure of 2,5-Hexanedione, CAS Number: 110-13-4
2,5-Hexanedione (CAS 110-13-4)
Alternate Names:
Acetonylacetone
Application:
2,5-Hexanedione is a useful starting reagent for diels-alder cycloaddition reactions
CAS Number:
110-13-4
Molecular Weight:
114.14
Molecular Formula:
C6H10O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,5-Hexanedione is a neurotoxic metabolite of n-hexanes and methyl n-butyl ketone. 2,5-Hexanedione is also used as the starting material for diels-alder cycloaddition reactions, such as a reactions with amines to form 2,5-dimethylpyrroles. 2,5-Hexanedione is a ketone compound that is obtained from acetone. It serves as a catalyst in the production of polymeric materials. Moreover, it finds application as a reagent in the synthesis of polysaccharides.
2,5-Hexanedione (CAS 110-13-4) References
- 2,5-hexanedione-induced testicular injury. | Boekelheide, K., et al. 2003. Annu Rev Pharmacol Toxicol. 43: 125-47. PMID: 12471174
- 2,5-hexanedione altered the degradation of low-molecular-weight neurofilament in rat nerve tissues. | Song, F., et al. 2012. Food Chem Toxicol. 50: 4277-84. PMID: 22967723
- Effects of 2,5-hexanedione on angiogenesis and vasculogenesis in chick embryos. | Cheng, X., et al. 2015. Reprod Toxicol. 51: 79-89. PMID: 25549948
- 2,5-hexanedione induced apoptosis of rat bone marrow mesenchymal stem cells by reactive oxygen species. | Liu, S., et al. 2016. J Occup Health. 58: 170-8. PMID: 27010086
- 2,5-hexanedione downregulates nerve growth factor and induces neuron apoptosis in the spinal cord of rats via inhibition of the PI3K/Akt signaling pathway. | Wang, Z., et al. 2017. PLoS One. 12: e0179388. PMID: 28654704
- 2,5-Hexanedione induces dopaminergic neurodegeneration through integrin αMβ2/NADPH oxidase axis-mediated microglial activation. | Zhang, C., et al. 2018. Cell Death Dis. 9: 60. PMID: 29352205
- 2,5-Hexanedione increases the percentage of proliferative Sox2+ cells in rat hippocampus. | Wang, L., et al. 2018. Toxicol Ind Health. 34: 589-595. PMID: 29764313
- Proapoptotic effects of 2,5‑hexanedione on pheochromocytoma cells via oxidative injury. | Qi, B., et al. 2019. Mol Med Rep. 20: 3249-3255. PMID: 31432125
- 2,5-hexanedione-induced deregulation of axon-related microRNA expression in rat nerve tissues. | Piao, F., et al. 2020. Toxicol Lett. 320: 95-102. PMID: 31760062
- 2,5-Hexanedione influences primordial follicular development in cultured neonatal mouse ovaries by interfering with the PI3K signaling pathway via miR-214-3p. | Zeng, J., et al. 2020. Toxicol Appl Pharmacol. 409: 115335. PMID: 33197454
- 2,5-hexanedione induces NLRP3 inflammasome activation and neurotoxicity through NADPH oxidase-dependent pathway. | Huang, R., et al. 2021. Free Radic Biol Med. 162: 561-570. PMID: 33212186
- 2,5-Hexanedione induced apoptosis in rat spinal cord neurons and VSC4.1 cells via the proNGF/p75NTR and JNK pathways. | Luo, M., et al. 2021. Biosci Rep. 41: PMID: 33792642
- Iodine-Modified Pd Catalysts Promote the Bifunctional Catalytic Synthesis of 2,5-Hexanedione from C6 Furan Aldehydes. | Tong, Z., et al. 2022. ChemSusChem. 15: e202102444. PMID: 34918485
- 2,5-Hexanedione Affects Ovarian Granulosa Cells in Swine by Regulating the CDKN1A Gene: A Transcriptome Analysis. | Chen, Y., et al. 2023. Vet Sci. 10: PMID: 36977240
- One-Pot Conversion of Cellulose into 2,5-Hexanedione in H2O-Tetrahydrofuran Co-Solvents. | Shi, N., et al. 2023. ACS Omega. 8: 11574-11582. PMID: 37008153
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Hexanedione, 100 g | sc-238390 | 100 g | $122.00 |
