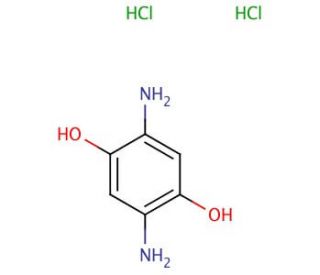
Molecular structure of 2,5-Diaminohydroquinone dihydrochloride, CAS Number: 24171-03-7
2,5-Diaminohydroquinone dihydrochloride (CAS 24171-03-7)
Application:
2,5-Diaminohydroquinone dihydrochloride is used in the preparation of polymers and synthetically cross-linked polyimide and silica hybrid films
CAS Number:
24171-03-7
Purity:
≥90%
Molecular Weight:
213.06
Molecular Formula:
C6H8N2O2•2HCl
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,5-Diaminohydroquinone dihydrochloride is a derivative of the aromatic amine, 2,5-diaminobenzene-1,4-diol (DABD). DABD is a useful compound in organic synthesis, as it can be used as a building block for the preparation of a variety of heterocyclic compounds. 2,5-Diaminohydroquinone dihydrochloride is used in the preparation of polymers and synthetically cross-linked polyimide and silica hybrid films. This compound acts as a nucleophilic catalyst in organic reactions. Studies have found that it can affect the metabolism of cells, as it may act to increase the activity of certain enzymes.
2,5-Diaminohydroquinone dihydrochloride (CAS 24171-03-7) References
- Stepwise formation of a molecular square with bridging NH,O-substituted dicarbene building blocks. | Conrady, FM., et al. 2011. J Am Chem Soc. 133: 11496-9. PMID: 21728329
- Electronic Conductivity, Ferrimagnetic Ordering, and Reductive Insertion Mediated by Organic Mixed-Valence in a Ferric Semiquinoid Metal-Organic Framework. | Darago, LE., et al. 2015. J Am Chem Soc. 137: 15703-11. PMID: 26573183
- Conversion of Imine to Oxazole and Thiazole Linkages in Covalent Organic Frameworks. | Waller, PJ., et al. 2018. J Am Chem Soc. 140: 9099-9103. PMID: 29999317
- A H-bond stabilized quinone electrode material for Li-organic batteries: the strength of weak bonds. | Sieuw, L., et al. 2019. Chem Sci. 10: 418-426. PMID: 30746090
- A Hollow Microtubular Triazine- and Benzobisoxazole-Based Covalent Organic Framework Presenting Sponge-Like Shells That Functions as a High-Performance Supercapacitor. | El-Mahdy, AFM., et al. 2019. Chem Asian J. 14: 1429-1435. PMID: 30817093
- Zinc ion interactions in a two-dimensional covalent organic framework based aqueous zinc ion battery. | Khayum M, A., et al. 2019. Chem Sci. 10: 8889-8894. PMID: 31762974
- Screen printing directed synthesis of covalent organic framework membranes with water sieving property. | Li, J., et al. 2020. Chem Commun (Camb). 56: 6519-6522. PMID: 32458910
- New Polyporphyrin Arrays with Controlled Fluorescence Obtained by Diaxial Sn(IV)-Porphyrin Phenolates Chelation with Cu2+ Cation. | Mamardashvili, GM., et al. 2021. Polymers (Basel). 13: PMID: 33800405
- Applications of Dynamic Covalent Chemistry Concept towards Tailored Covalent Organic Framework Nanomaterials: A Review. | Hu, J., et al. 2020. ACS Appl Nano Mater. 3: 6239-6269. PMID: 34327307
- Postsynthetic functionalization of covalent organic frameworks. | Yusran, Y., et al. 2020. Natl Sci Rev. 7: 170-190. PMID: 34692030
- High-Strength, Microporous, Two-Dimensional Polymer Thin Films with Rigid Benzoxazole Linkage. | Miller, KA., et al. 2022. ACS Appl Mater Interfaces. 14: 1861-1873. PMID: 34978172
- Stabilizing a zinc anode via a tunable covalent organic framework-based solid electrolyte interphase. | Aupama, V., et al. 2023. Nanoscale. 15: 9003-9013. PMID: 37128979
- New Naphthalene-Based Polyimide as an Environment-Friendly Organic Cathode Material for Lithium Batteries | and Alexander V. Mumyatov, Alexander F. Shestakov, Nadezhda N. Dremova, Keith J. Stevenson, Pavel A. Troshin. May 2019. Energy Technology. Volume7, Issue5: 1801016.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Diaminohydroquinone dihydrochloride, 1 g | sc-231066 | 1 g | $179.00 |
