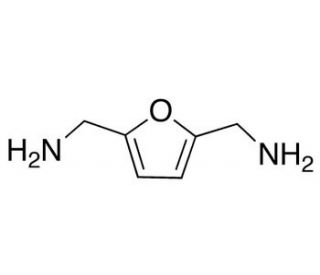
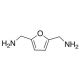
2,5-Bis(aminomethyl)furan (CAS 2213-51-6)
QUICK LINKS
2,5-Bis(aminomethyl)furan is a versatile organic compound, featuring a furan ring substituted with aminomethyl groups at the 2 and 5 positions, which plays a pivotal role in various research domains, particularly in the synthesis of polymers, materials science, and as an intermediate in organic synthesis. Its unique structure facilitates the formation of novel polymers and materials with enhanced properties, such as increased thermal stability, conductivity, and biodegradability, making it a compound of interest in the development of sustainable materials. Furthermore, 2,5-Bis(aminomethyl)furan serves as a critical intermediate in the synthesis of more complex organic molecules, acting as a building block in the construction of a wide array of compounds due to its reactive aminomethyl groups. These groups offer points of functionalization, enabling the introduction of various side chains and functionalities, which are instrumental in the tailor-made synthesis of molecules for specific research applications. This compound′s versatility and reactivity thus underscore its importance in advancing research in materials science and organic chemistry, providing a foundation for the development of innovative materials and the exploration of new realms of chemical synthesis.
2,5-Bis(aminomethyl)furan (CAS 2213-51-6) References
- Facile Chemical Access to Biologically Active Norcantharidin Derivatives from Biomass. | Galkin, KI., et al. 2017. Molecules. 22: PMID: 29231880
- The Increasing Value of Biomass: Moving From C6 Carbohydrates to Multifunctionalized Building Blocks via 5-(hydroxymethyl)furfural. | Galkin, KI. and Ananikov, VP. 2020. ChemistryOpen. 9: 1135-1148. PMID: 33204585
- One-Step Reductive Amination of 5-Hydroxymethylfurfural into 2,5-Bis(aminomethyl)furan over Raney Ni. | Wei, Z., et al. 2021. ChemSusChem. 14: 2308-2312. PMID: 33909345
- Reductive Amination of 5-Hydroxymethylfurfural to 2,5-Bis(aminomethyl)furan over Alumina-Supported Ni-Based Catalytic Systems. | Wei, Z., et al. 2022. ChemSusChem. 15: e202200233. PMID: 35225422
- Highly selective synthesis of 2,5-bis(aminomethyl)furan via catalytic amination of 5-(hydroxymethyl)furfural with NH3 over a bifunctional catalyst. | Yuan, H., et al. 2019. RSC Adv. 9: 38877-38881. PMID: 35540204
- Sustainable Approaches to Selective Conversion of Cellulose Into 5-Hydroxymethylfurfural Promoted by Heterogeneous Acid Catalysts: A Review. | Yao, Y., et al. 2022. Front Chem. 10: 880603. PMID: 35620654
- Brand-new Biomass-based Vinyl Polymers from 5-Hydroxymethylfurfural | , et al. (2008). Polymer Journal. volume 40,: pages 1164–1169.
- Synthesis of 2,5-diformylfuran from renewable carbohydrates and its applications: A review | J Dai - Green Energy & Environment, 2021 - Elsevier. February 2021,. Green Energy & Environment. Volume 6, Issue 1,: Pages 22-32.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Bis(aminomethyl)furan, 25 mg | sc-209343 | 25 mg | $286.00 | |||
2,5-Bis(aminomethyl)furan, 250 mg | sc-209343A | 250 mg | $2399.00 |
