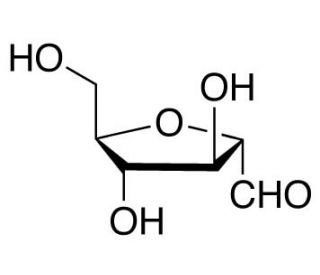
Molecular structure of 2,5-Anhydro-D-mannose, CAS Number: 495-75-0
2,5-Anhydro-D-mannose (CAS 495-75-0)
Alternate Names:
Chitose; D-2,5-Anhydromannose
Application:
2,5-Anhydro-D-mannose is a mixture of mannose acetal and aldehyde forms
CAS Number:
495-75-0
Molecular Weight:
162.14
Molecular Formula:
C6H10O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,5-Anhydro-D-mannose is a mixture of the acetal and aldehyde forms. 2,5-Anhydro-D-mannose is a monosaccharide, which is a type of sugar molecule. It is structurally similar to D-mannose but contains an additional anhydro (dehydration) group between the second and fifth carbon atoms. In research, 2,5-Anhydro-D-mannose is primarily used as a precursor or building block for the synthesis of alginate-based materials. It serves as a starting material for the production of alginate hydrogels, microcapsules, and scaffolds.
2,5-Anhydro-D-mannose (CAS 495-75-0) References
- Use of chromium-adenosine triphosphate and lyxose to elucidate the kinetic mechanism and coordination state of the nucleotide substrate for yeast hexokinase. | Danenberg, KD. and Cleland, WW. 1975. Biochemistry. 14: 28-39. PMID: 1089014
- Hydrazinolysis and nitrous acid deamination of the carbohydrate moiety of alpha1-acid glycoprotein. | Bayard, B. and Fournet, B. 1976. Carbohydr Res. 46: 75-86. PMID: 1248015
- Product individuality of commercially available low-molecular-weight heparins and their generic versions: therapeutic implications. | Maddineni, J., et al. 2006. Clin Appl Thromb Hemost. 12: 267-76. PMID: 16959680
- Bovine liver fructokinase: purification and kinetic properties. | Raushel, FM. and Cleland, WW. 1977. Biochemistry. 16: 2169-75. PMID: 193556
- Changes in the structure and biological property of N----O sulfate-transferred, N-resulfated heparin. | Uchiyama, H. and Nagasawa, K. 1991. J Biol Chem. 266: 6756-60. PMID: 2016290
- Monoclonal antibodies specific for oligosaccharides prepared by partial nitrous acid deamination of heparin. | Pejler, G., et al. 1988. J Biol Chem. 263: 5197-201. PMID: 2451664
- Extraction of chitosan and its oligomers from shrimp shell waste, their characterization and antimicrobial effect. | Varun, TK., et al. 2017. Vet World. 10: 170-175. PMID: 28344399
- Examining the effect of common nitrosating agents on chitosan using a glucosamine oligosaccharide model system. | Allison, CL., et al. 2019. Carbohydr Polym. 203: 285-291. PMID: 30318215
- Influence of Preparation Methods of Chitooligosaccharides on Their Physicochemical Properties and Their Anti-Inflammatory Effects in Mice and in RAW264.7 Macrophages. | Sánchez, Á., et al. 2018. Mar Drugs. 16: PMID: 30400250
- Identification of low molecular weight degradation products from chitin and chitosan by electrospray ionization time-of-flight mass spectrometry. | Allison, CL., et al. 2020. Carbohydr Res. 493: 108046. PMID: 32497941
- 2,5-Anhydro-d-Mannose End-Functionalized Chitin Oligomers Activated by Dioxyamines or Dihydrazides as Precursors of Diblock Oligosaccharides. | Mo, IV., et al. 2020. Biomacromolecules. 21: 2884-2895. PMID: 32539358
- Production of Structurally Defined Chito-Oligosaccharides with a Single N-Acetylation at Their Reducing End Using a Newly Discovered Chitinase from Paenibacillus pabuli. | Li, J., et al. 2021. J Agric Food Chem. 69: 3371-3379. PMID: 33688734
- A novel method for determining rate constants for dehydration of aldehyde hydrates. | Rendina, AR., et al. 1984. Biochemistry. 23: 5148-56. PMID: 6095890
- Evidence for a 3-O-sulfated D-glucosamine residue in the antithrombin-binding sequence of heparin. | Lindahl, U., et al. 1980. Proc Natl Acad Sci U S A. 77: 6551-5. PMID: 6935668
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Anhydro-D-mannose, 10 mg | sc-220809 | 10 mg | $398.00 |
