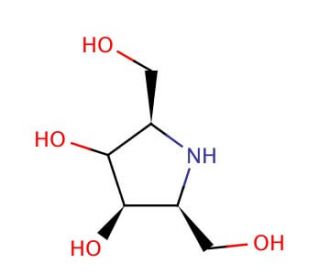
2,5-Anhydro-2,5-imino-D-glucitol (CAS 132295-44-4)
QUICK LINKS
2,5-Anhydro-2,5-imino-D-glucitol, a potent glycomimetic compound, has garnered significant attention in scientific research for its unique structural properties and diverse applications in glycobiology studies. Its mechanism of action centers around its ability to mimic the structural features of natural carbohydrates, particularly in glycan recognition processes. This compound acts as a structural analog of glucose, effectively engaging with carbohydrate-binding proteins and receptors, thus modulating various cellular signaling pathways involved in cell adhesion, migration, and differentiation. Researchers have utilized its glycomimetic properties to dissect the molecular mechanisms underlying carbohydrate-protein interactions, elucidating the roles of glycans in fundamental cellular processes and disease pathogenesis. Moreover, this compound has been instrumental in the development of glycan-based biomaterials for tissue engineering applications, facilitating the design of bioactive scaffolds and drug delivery systems with enhanced biocompatibility and targeting capabilities. Furthermore, its utility extends to the synthesis of glycomimetic libraries for high-throughput screening assays, enabling the discovery of novel glycan-binding molecules and the characterization of glycan-mediated host-pathogen interactions. Through its versatile applications, 2,5-Anhydro-2,5-imino-D-glucitol continues to advance our understanding of glycan biology and pave the way for innovative research strategies aimed at unraveling the complex roles of glycans in health and disease.
2,5-Anhydro-2,5-imino-D-glucitol (CAS 132295-44-4) References
- Isofagomine- and 2,5-anhydro-2,5-imino-D-glucitol-based glucocerebrosidase pharmacological chaperones for Gaucher disease intervention. | Yu, Z., et al. 2007. J Med Chem. 50: 94-100. PMID: 17201413
- Protecting-group-free synthesis of 2-deoxy-aza-sugars. | Dangerfield, EM., et al. 2009. Molecules. 14: 5298-307. PMID: 20032893
- Potent aminocyclitol glucocerebrosidase inhibitors are subnanomolar pharmacological chaperones for treating gaucher disease. | Trapero, A., et al. 2012. J Med Chem. 55: 4479-88. PMID: 22512696
- The lipophilic bullet hits the targets: medicinal chemistry of adamantane derivatives. | Wanka, L., et al. 2013. Chem Rev. 113: 3516-604. PMID: 23432396
- Expanding role of molecular chaperones in regulating α-synuclein misfolding; implications in Parkinson's disease. | Sharma, SK. and Priya, S. 2017. Cell Mol Life Sci. 74: 617-629. PMID: 27522545
- Studies on the effect of glycoprotein processing inhibitors on fusion of L6 myoblast cell lines. | Spearman, MA., et al. 1987. Exp Cell Res. 168: 116-26. PMID: 2946596
- Inhibitors of the biosynthesis and processing of N-linked oligosaccharide chains. | Elbein, AD. 1987. Annu Rev Biochem. 56: 497-534. PMID: 3304143
- Synthesis and alpha-D-glucosidase inhibitory activity of N-substituted valiolamine derivatives as potential oral antidiabetic agents. | Horii, S., et al. 1986. J Med Chem. 29: 1038-46. PMID: 3519969
- New entry to pyrrolidine homoazasugars: conversion of d-arabinose into 2,5-anhydro-2,5-imino-d-glucitol via aminohomologation | Alessandro Dondoni and, Daniela Perrone. 1999. Tetrahedron Letters. 40: 9375-9378.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,5-Anhydro-2,5-imino-D-glucitol, 10 mg | sc-220802 | 10 mg | $430.00 |
