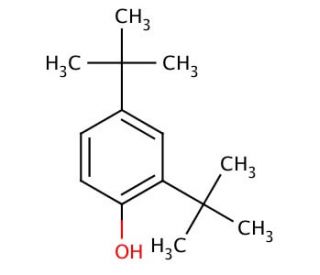
2,4-Di-tert-butylphenol (CAS 96-76-4)
QUICK LINKS
2,4-Di-tert-butylphenol was used to study its alkylation reaction with cinnamyl alcohol in the presence of Al-substituted disordered hexagonal molecular silica catalysts. Another interesting property of 2,4-Di-tert-butylphenol is that it possess antioxidative activity. 2,4-Di-tert-butylphenol (2,4-DTBP) is a phenolic compound characterized by the presence of two tert-butyl groups linked to its phenolic hydroxyl group. This compound serves as a intermediary in the creation of diverse organic compounds and serves as a foundational element in the production of organic substances. Furthermore, 2,4-Di-tert-butylphenol plays a key role in the formulation of plasticizers, rubber additives, and other industrial goods. An understanding of both the configuration and attributes of 2,4-Di-tert-butylphenol holds significance in comprehending its wide-ranging applications across multiple sectors. In the realm of organic compound synthesis, 2,4-Di-tert-butylphenol serves as an initial building block. Additionally, its utility extends to the generation of plasticizers and various industrial commodities. Noteworthy recent investigations have highlighted the potential of 2,4-Di-tert-butylphenol in producing chiral compounds, pivotal in drug and compound synthesis. Moreover, this compound has demonstrated its efficacy in polymer synthesis, offering versatile applications in numerous fields.
2,4-Di-tert-butylphenol (CAS 96-76-4) References
- 2,4-Di-tert-butylphenol from sweet potato protects against oxidative stress in PC12 cells and in mice. | Choi, SJ., et al. 2013. J Med Food. 16: 977-83. PMID: 24074359
- 2,4‑Di‑tert‑butylphenol, a potential HDAC6 inhibitor, induces senescence and mitotic catastrophe in human gastric adenocarcinoma AGS cells. | Song, YW., et al. 2018. Biochim Biophys Acta Mol Cell Res. 1865: 675-683. PMID: 29427610
- Impact of preoxidation of UV/persulfate on disinfection byproducts by chlorination of 2,4-Di-tert-butylphenol. | Wang, Q., et al. 2018. J Hazard Mater. 358: 450-458. PMID: 30036772
- Removal of bisphenol A and 2,4-Di-tert-butylphenol from landfill leachate using plant- based coagulant. | Aziz, A., et al. 2018. Waste Manag Res. 36: 975-984. PMID: 30058954
- Anti-inflammatory and anticancer activities of erythrodiol-3-acetate and 2,4-di-tert-butylphenol isolated from Humboldtia unijuga. | Nair, RVR., et al. 2020. Nat Prod Res. 34: 2319-2322. PMID: 30475646
- Unexpectedly high concentrations of 2,4-di-tert-butylphenol in human urine. | Liu, R. and Mabury, SA. 2019. Environ Pollut. 252: 1423-1428. PMID: 31265952
- Natural Sources and Bioactivities of 2,4-Di-Tert-Butylphenol and Its Analogs. | Zhao, F., et al. 2020. Toxins (Basel). 12: PMID: 31935944
- Organic acids and 2,4-Di-tert-butylphenol: major compounds of Weissella confusa WM36 cell-free supernatant against growth, survival and virulence of Salmonella Typhi. | Pelyuntha, W., et al. 2020. PeerJ. 8: e8410. PMID: 31998561
- 2,4-Di-Tert-Butylphenol Isolated From an Endophytic Fungus, Daldinia eschscholtzii, Reduces Virulence and Quorum Sensing in Pseudomonas aeruginosa. | Mishra, R., et al. 2020. Front Microbiol. 11: 1668. PMID: 32849344
- Butylated hydroxyl-toluene, 2,4-Di-tert-butylphenol, and phytol of Chlorella sp. protect the PC12 cell line against H2O2-induced neurotoxicity. | Vahdati, SN., et al. 2022. Biomed Pharmacother. 145: 112415. PMID: 34775236
- 2,4-di-tert-butylphenol exposure impairs osteogenic differentiation. | Dương, TB., et al. 2023. Toxicol Appl Pharmacol. 461: 116386. PMID: 36682590
- 2,4-Di-tert-butylphenol Induces Adipogenesis in Human Mesenchymal Stem Cells by Activating Retinoid X Receptors. | Ren, XM., et al. 2023. Endocrinology. 164: PMID: 36750942
- A Novel Finding: 2,4-Di-tert-butylphenol from Streptomyces bacillaris ANS2 Effective Against Mycobacterium tuberculosis and Cancer Cell Lines. | Kaari, M., et al. 2023. Appl Biochem Biotechnol.. PMID: 36881320
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,4-Di-tert-butylphenol, 5 g | sc-238312 | 5 g | $20.00 | |||
2,4-Di-tert-butylphenol, 100 g | sc-238312A | 100 g | $37.00 |
