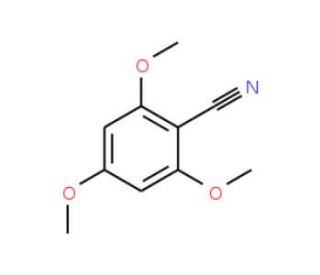
2,4,6-Trimethoxybenzonitrile (CAS 2571-54-2)
QUICK LINKS
2,4,6-Trimethoxybenzonitrile (TMBN) is a colorless solid with remarkable potential applications in both research and industry. This aromatic nitrile compound exhibits solubility in organic solvents and displays stability at room temperature. The potential applications of 2,4,6-Trimethoxybenzonitrile in scientific research span a wide range. It serves as a fundamental starting material for synthesizing an assortment of compounds. As an aromatic nitrile compound, 2,4,6-Trimethoxybenzonitrile showcases a diverse array of potential reactions. It readily partakes in nucleophilic addition reactions, nucleophilic substitution reactions, and electrophilic substitution reactions. Furthermore, it displays versatility by engaging in other reactions, such as hydrolysis, oxidation, and reduction.
2,4,6-Trimethoxybenzonitrile (CAS 2571-54-2) References
- Gold(I) complexes with hydrogen-bond supported heterocyclic carbenes as active catalysts in reactions of 1,6-enynes. | Bartolomé, C., et al. 2008. Inorg Chem. 47: 11391-7. PMID: 18947178
- Asymmetric synthesis of axially chiral 1-aryl-5,6,7,8-tetrahydroquinolines by cobalt-catalyzed [2 + 2 + 2] cycloaddition reaction of 1-aryl-1,7-octadiynes and nitriles. | Hapke, M., et al. 2010. J Org Chem. 75: 3993-4003. PMID: 20481604
- Cationic gallium(III) halide complexes: a new generation of π-Lewis acids. | Tang, S., et al. 2012. Chemistry. 18: 10239-43. PMID: 22674541
- Pd(OAc)2-catalyzed alkoxylation of arylnitriles via sp2 C-H bond activation using cyano as the directing group. | Li, W. and Sun, P. 2012. J Org Chem. 77: 8362-6. PMID: 22946882
- A hexanuclear gold cluster supported by three-center-two-electron bonds and aurophilic interactions. | Smirnova, ES. and Echavarren, AM. 2013. Angew Chem Int Ed Engl. 52: 9023-6. PMID: 23857851
- Bioherbicidal Activity and Metabolic Profiling of Potent Allelopathic Plant Fractions Against Major Weeds of Wheat-Way Forward to Lower the Risk of Synthetic Herbicides. | Anwar, S., et al. 2021. Front Plant Sci. 12: 632390. PMID: 34567017
- Dual Nickel/Photoredox-Catalyzed Deaminative Cross-Coupling of Sterically Hindered Primary Amines. | Dorsheimer, JR., et al. 2021. J Am Chem Soc. 143: 19294-19299. PMID: 34767360
- Photocatalytic C(sp3) radical generation via C-H, C-C, and C-X bond cleavage. | Huang, CY., et al. 2022. Chem Sci. 13: 5465-5504. PMID: 35694342
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,4,6-Trimethoxybenzonitrile, 5 g | sc-225719 | 5 g | $58.00 |
