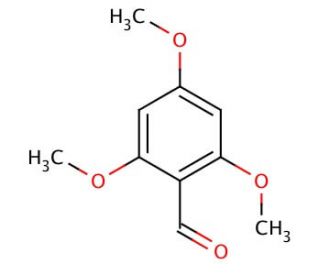
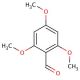
2,4,6-Trimethoxybenzaldehyde (CAS 830-79-5)
QUICK LINKS
2,4,6-Trimethoxybenzaldehyde (TMB) is a natural compound present in various plants, including the cinnamon tree′s bark and the camphor tree′s leaves. Its significance lies in being a vital intermediary in the synthesis of flavonoids, phenolic acids, and other biologically active compounds. Consequently, 2,4,6-Trimethoxybenzaldehyde has garnered considerable attention in recent times for its promising applications in medical research. While the exact workings of 2,4,6-Trimethoxybenzaldehyde remain partially veiled, scientists speculate on its mechanisms of action. It is thought that this compound operates by inhibiting the production of inflammatory mediators like prostaglandins and leukotrienes, which contribute to inflammation. Additionally, 2,4,6-Trimethoxybenzaldehyde may also function as an antioxidant, effectively scavenging free radicals and safeguarding cells against oxidative damage. 2,4,6-Trimethoxybenzaldehyde holds great promise in the realm of medicinal research due to its natural origin, and versatile applications. However, further research is needed to fully comprehend its complete mechanism of action and unlock its full potential.
2,4,6-Trimethoxybenzaldehyde (CAS 830-79-5) References
- Synthesis and biological evaluation of methoxyphenyl porphyrin derivatives as potential photodynamic agents. | Milanesio, ME., et al. 2001. Bioorg Med Chem. 9: 1943-9. PMID: 11504630
- The ortho backbone amide linker (o-BAL) is an easily prepared and highly acid-labile handle for solid-phase synthesis. | Boas, U., et al. 2002. J Comb Chem. 4: 223-8. PMID: 12005482
- Stimuli-responsive supramolecular assemblies of linear-dendritic copolymers. | Gillies, ER., et al. 2004. J Am Chem Soc. 126: 11936-43. PMID: 15382929
- Methoxy-phenyl substituted ansa-titanocenes as potential anti-cancer drugs derived from fulvenes and titanium dichloride. | Tacke, M., et al. 2004. J Inorg Biochem. 98: 1987-94. PMID: 15541486
- pH-Responsive copolymer assemblies for controlled release of doxorubicin. | Gillies, ER. and Fréchet, JM. 2005. Bioconjug Chem. 16: 361-8. PMID: 15769090
- Regioselective synthesis and estrogenicity of (+/-)-8-alkyl-5,7-dihydroxy-4-(4-hydroxyphenyl)-3,4-dihydrocoumarins. | Roelens, F., et al. 2005. Eur J Med Chem. 40: 1042-51. PMID: 15951062
- Expansile nanoparticles: synthesis, characterization, and in vivo efficacy of an acid-responsive polymeric drug delivery system. | Griset, AP., et al. 2009. J Am Chem Soc. 131: 2469-71. PMID: 19182897
- pH-responsive biodegradable micelles based on acid-labile polycarbonate hydrophobe: synthesis and triggered drug release. | Chen, W., et al. 2009. Biomacromolecules. 10: 1727-35. PMID: 19469499
- pH-Sensitive degradable polymersomes for triggered release of anticancer drugs: a comparative study with micelles. | Chen, W., et al. 2010. J Control Release. 142: 40-6. PMID: 19804803
- Discovery of a clinical stage multi-kinase inhibitor sodium (E)-2-{2-methoxy-5-[(2',4',6'-trimethoxystyrylsulfonyl)methyl]phenylamino}acetate (ON 01910.Na): synthesis, structure-activity relationship, and biological activity. | Reddy, MV., et al. 2011. J Med Chem. 54: 6254-76. PMID: 21812421
- Stabilizing the imidazolium cation in hydroxide-exchange membranes for fuel cells. | Wang, J., et al. 2013. ChemSusChem. 6: 2079-82. PMID: 24039129
- Acid-Triggered Nanoexpansion Polymeric Micelles for Enhanced Photodynamic Therapy. | Zhong, S., et al. 2019. ACS Appl Mater Interfaces. 11: 33697-33705. PMID: 31487149
- Synthesis, Antimicrobial and Antioxidant Activities of 2-Isoxazoline Derivatives. | Alshamari, A., et al. 2020. Molecules. 25: PMID: 32961855
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,4,6-Trimethoxybenzaldehyde, 10 g | sc-238365 | 10 g | $51.00 |
