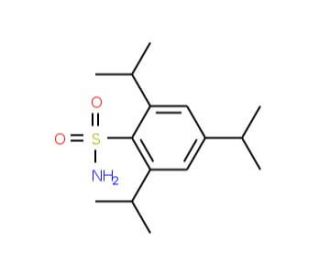
2,4,6-Triisopropylbenzenesulfonamide (CAS 105536-22-9)
QUICK LINKS
2,4,6-Triisopropylbenzenesulfonamide is a chemical used primarily in the field of organic synthesis as a protecting group for amines. In synthetic chemistry research, it is applied to prevent the reactivity of amine functional groups during complex chemical reactions, allowing for selective modifications of other parts of the molecule. Beyond its use as a protecting group, this compound also serves as a reagent in the study of sulfonamide chemistry, exploring its stability, reactivity, and subsequent deprotection under various conditions. Researchers utilize 2,4,6-Triisopropylbenzenesulfonamide to develop new synthetic methodologies that may enhance the efficiency and selectivity of chemical transformations. It is also of interest in materials science, where its incorporation into polymers is investigated for the potential to impart specific physical or chemical properties.
2,4,6-Triisopropylbenzenesulfonamide (CAS 105536-22-9) References
- Targeting of single-stranded DNA and RNA containing adjacent pyrimidine and purine tracts by triple helix formation with circular and clamp oligonucleotides. | Maksimenko, AV., et al. 2000. Eur J Biochem. 267: 3592-603. PMID: 10848976
- 2,4,6-Triisopropylbenzenesulfonamide: Monte Carlo structure solution from X-ray powder diffraction data for a molecular system containing four independent asymmetric rotors. | Tremayne, M., et al. 1999. Acta Crystallogr B. 55: 1068-1074. PMID: 10927448
- Direct Space Structure Solution Applications. | Tremayne, M. 2004. J Res Natl Inst Stand Technol. 109: 49-63. PMID: 27366596
- Manganese-Catalyzed N-Alkylation of Sulfonamides Using Alcohols. | Reed-Berendt, BG. and Morrill, LC. 2019. J Org Chem. 84: 3715-3724. PMID: 30789263
- Oxidative N-Heterocyclic Carbene Catalysis. | De Risi, C., et al. 2023. Chemistry. 29: e202202467. PMID: 36205918
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,4,6-Triisopropylbenzenesulfonamide, 5 g | sc-266116 | 5 g | $106.00 |
