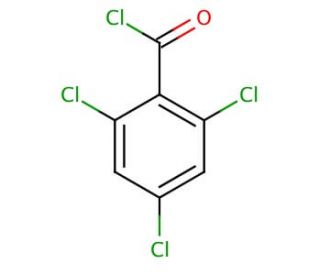
2,4,6-Trichlorobenzoyl chloride (CAS 4136-95-2)
QUICK LINKS
2,4,6-Trichlorobenzoyl chloride is a chemical compound that functions as an acylating agent in organic synthesis. It is used to introduce the 2,4,6-trichlorobenzoyl group into various organic molecules, allowing for the modification of their chemical properties. Trimethylolpropane tris(3-mercaptopropionate) ′s mode of action involves reacting with nucleophilic functional groups, such as hydroxyl or amino groups, to form acyl derivatives. Through this process, 2,4,6-Trichlorobenzoyl chloride facilitates the creation of new compounds with altered reactivity and properties, enabling explore the effects of these modifications in experimental applications. Its acylating properties make it a useful for the development of novel organic molecules and materials, contributing to the advancement of chemical innovation.
2,4,6-Trichlorobenzoyl chloride (CAS 4136-95-2) References
- Synthesis of γ- and δ-lactone natural products by employing a trans-cis isomerization/lactonization strategy. | Ono, M., et al. 2013. Chem Pharm Bull (Tokyo). 61: 464-70. PMID: 23546007
- Novel synthetic method for the preparation of amphiphilic hyaluronan by means of aliphatic aromatic anhydrides. | Huerta-Angeles, G., et al. 2014. Carbohydr Polym. 111: 883-91. PMID: 25037428
- New seco-limonoids from Cipadessa baccifera: Isolation, structure determination, synthesis and their antiproliferative activities. | Siva, B., et al. 2017. Fitoterapia. 117: 34-40. PMID: 28065696
- (E)-Ethyl-2-cyano-2-(((2,4,6-trichlorobenzoyl)oxy)imino)acetate: A Modified Yamaguchi Reagent for Enantioselective Esterification, Thioesterification, Amidation, and Peptide Synthesis. | Chandra, J., et al. 2018. ACS Omega. 3: 6120-6133. PMID: 30023940
- Synthesis of erythronolide a via a very efficient macrolactonization under usual acylation conditions with the Yamaguchi reagent | Masataka Hikota, Youji Sakurai, Kiyoshi Horita, Osamu Yonemitsu ∗. 1990. Tetrahedron Letters. 31: 6367-6370.
- Improved preparation of angelate esters | Benoît Hartmann, Alice M. Kanazawa, Jean-Pierre Deprés, Andrew E. Greene ∗. 1991. Tetrahedron Letters. 32: 5077-5080.
- The total synthesis of macrosphelides A and E from carbohydrate precursors | G.V.M. Sharma, Ch.Chandra Mouli. 2002. Tetrahedron Letters. 43: 9159-9161.
- A total synthesis of macrosphelides C and F from l-(+)-arabinose | G.V.M. Sharma, Ch.Chandra Mouli. 2003. Tetrahedron Letters. 44: 8161-8163.
- A stereoselective synthesis of verbalactone-determination of absolute stereochemistry | G.V.M. Sharma, Ch. Govardhan Reddy. 2004. Tetrahedron Letters. 45: 7483-7485.
- δ-Lactone formation from δ-hydroxy-trans-α,β-unsaturated carboxylic acids accompanied by trans–cis isomerization: synthesis of (−)-tetra-O-acetylosmundalin | Machiko Ono a, Xi Ying Zhao b, Yuki Shida b, Hiroyuki Akita b. 2007. Tetrahedron. 63: 10140-10148.
- Synthesis, characterization, and antimicrobial activities of clubbed [1,2,4]-oxadiazoles with fluorobenzimidazoles | Ganesh R. Jadhav, Mohammad U. Shaikh, Rajesh P. Kale, Anand R. Ghawalkar, Charansingh H. Gill. 2009. Journal of Heterocyclic Chemistry. 46: 980-987.
- NHC-catalysed enantioselective intramolecular formal [4+ 2] cycloadditions using carboxylic acids as azolium enolate precursors | Nassilia Attaba, Andrew D. Smith. 2020. Tetrahedron. 76.
- Applications of Yamaguchi Method to Esterification and Macrolactonization in Total Synthesis of Bioactive Natural Products | Dr. Sasadhar Majhi. 2021. ChemistrySelect,. 6: 4178-4206.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,4,6-Trichlorobenzoyl chloride, 5 g | sc-251854 | 5 g | $101.00 |
