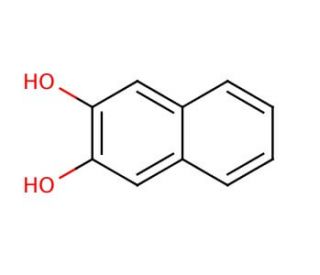
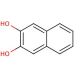
Molecular structure of 2,3-Dihydroxynaphthalene, CAS Number: 92-44-4
2,3-Dihydroxynaphthalene (CAS 92-44-4)
Alternate Names:
2,3-Naphthalenediol
CAS Number:
92-44-4
Molecular Weight:
160.17
Molecular Formula:
C10H8O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,3-Dihydroxynaphthalene is extensively utilized in organic chemistry, particularly in the synthesis of dyes and pigments. 2,3-Dihydroxynaphthalene serves as a key precursor in the production of various naphthoquinone derivatives, which are important for their application in creating colorfast and vibrant dyes applied in the textile industry. In analytical chemistry, 2,3-Dihydroxynaphthalene is employed in fluorescence studies due to its photophysical properties. 2,3-Dihydroxynaphthalene is employed in environmental chemistry for studying the degradation pathways and environmental fate of aromatic compounds that exhibit structural similarity to 2,3-Dihydroxynaphthalene.
2,3-Dihydroxynaphthalene (CAS 92-44-4) References
- Evidence for the existence of PAH-quinone reductase and catechol-O-methyltransferase in Mycobacterium vanbaalenii PYR-1. | Kim, YH., et al. 2004. J Ind Microbiol Biotechnol. 31: 507-16. PMID: 15549609
- Degradation of phenanthrene and anthracene by Nocardia otitidiscaviarum strain TSH1, a moderately thermophilic bacterium. | Zeinali, M., et al. 2008. J Appl Microbiol. 105: 398-406. PMID: 18312570
- Hydrolytic metal with a hydrophobic periphery: titanium(IV) complexes of naphthalene-2,3-diolate and interactions with serum albumin. | Tinoco, AD., et al. 2008. Inorg Chem. 47: 8380-90. PMID: 18710217
- The syntheses and characterizations of molybdenum(VI) complexes with catechol and 2,3-dihydroxynaphthalene, and the structure-effect relationship in their in vitro anticancer activities. | Feng, J., et al. 2012. Dalton Trans. 41: 8697-702. PMID: 22691976
- Synthesis and properties of butterfly-shaped expanded naphthofuran derivatives. | Nakanishi, K., et al. 2014. J Org Chem. 79: 2625-31. PMID: 24564354
- Titanium complex formation of organic ligands in titania gels. | Nishikiori, H., et al. 2015. Langmuir. 31: 964-9. PMID: 25535798
- Reversible Switching of Tb(III) Emission by Sensitization from 2,3-Dihydroxynaphthalene in an Isothermally Reversible Ionic Liquid. | Divya, KP. and Weiss, RG. 2015. J Phys Chem Lett. 6: 893-7. PMID: 26262669
- Stereochemistry and spectroscopic analysis of bis-Betti base derivatives of 2,3-dihydroxynaphthalene. | Zamani, M., et al. 2016. J Mol Model. 22: 86. PMID: 26994020
- The synthesis of novel chromogenic enzyme substrates for detection of bacterial glycosidases and their applications in diagnostic microbiology. | Burton, M., et al. 2018. Bioorg Med Chem. 26: 4841-4849. PMID: 30170924
- Organophosphorus decorated lithium borate and phosphate salts with extended π-conjugated backbone. | Delgado Rosario, E., et al. 2021. Dalton Trans. 50: 6667-6672. PMID: 33908542
- Wormlike micelles of CTAB with phenols and with the corresponding phenolate derivatives - When hydrophobicity and charge drive the coacervation. | Creatto, EJ., et al. 2022. J Colloid Interface Sci. 627: 355-366. PMID: 35863194
- 2,3-Dihydroxynaphthalene invoked surface oxygen vacancy effect on Fe2O3 nanorods for photoanodic signal transduction tactic. | Yan, M., et al. 2023. Biosens Bioelectron. 232: 115286. PMID: 37079991
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3-Dihydroxynaphthalene, 50 g | sc-238280 | 50 g | $49.00 |
