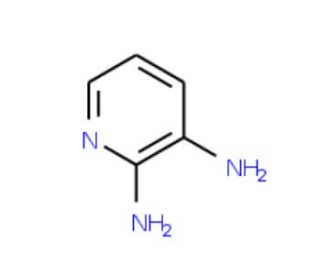
2,3-Diaminopyridine (CAS 452-58-4)
QUICK LINKS
2,3-Diaminopyridine is a chemical compound that functions as a nucleophilic reagent in various organic synthesis reactions. It acts as a building block in the formation of complex organic molecules, participating in the construction of heterocyclic compounds and intermediates. Its mechanism of action involves the activation of specific functional groups within the target molecule, facilitating the formation of new chemical bonds through nucleophilic substitution or addition reactions. 2,3-Diaminopyridine plays a role in the modification of organic molecules, enabling the introduction of amino groups into the molecular structure. Through its nucleophilic reactivity, 2,3-Diaminopyridine contributes to the diversification of chemical libraries and the synthesis of novel compounds with potential applications. Its ability to participate in selective chemical transformations may be useful for the development of new materials and the exploration of structure-activity relationships in organic chemistry.
2,3-Diaminopyridine (CAS 452-58-4) References
- Spectroscopic studies of the reaction of iodine with 2,3-diaminopyridine. | Al-Hashimi, NA. 2004. Spectrochim Acta A Mol Biomol Spectrosc. 60: 2181-4. PMID: 15249002
- 2,3-diaminopyridine bradykinin B1 receptor antagonists. | Kuduk, SD., et al. 2004. J Med Chem. 47: 6439-42. PMID: 15588075
- 2,3-Diaminopyridine as a platform for designing structurally unique nonpeptide bradykinin B1 receptor antagonists. | Feng, DM., et al. 2005. Bioorg Med Chem Lett. 15: 2385-8. PMID: 15837330
- Bioactivation of 2,3-diaminopyridine-containing bradykinin B1 receptor antagonists: irreversible binding to liver microsomal proteins and formation of glutathione conjugates. | Tang, C., et al. 2005. Chem Res Toxicol. 18: 934-45. PMID: 15962928
- Bradykinin B1 antagonists: SAR studies in the 2,3-diaminopyridine series. | Kuduk, SD., et al. 2005. Bioorg Med Chem Lett. 15: 3925-9. PMID: 15993596
- Cyclopropylamino acid amide as a pharmacophoric replacement for 2,3-diaminopyridine. Application to the design of novel bradykinin B1 receptor antagonists. | Wood, MR., et al. 2006. J Med Chem. 49: 1231-4. PMID: 16480259
- Studies on the synthesis, characterization, binding with DNA and activities of two cis-planaramineplatinum(II) complexes of the form: cis-PtL(NH3)Cl2 where L = 3-hydroxypyridine and 2,3-diaminopyridine. | Abdullah, A., et al. 2006. BMC Chem Biol. 6: 3. PMID: 16533399
- Experimental and theoretical studies on the functionalization reactions of 4-benzoyl-1,5-diphenyl-1H-pyrazole-3-carboxylic acid and acid chloride with 2,3-diaminopyridine. | Yildirim, I., et al. 2005. Molecules. 10: 559-71. PMID: 18007329
- Investigation of voltammetric enzyme-linked immunoassay system based on N-heterocyclic substrate of 2,3-diaminopyridine. | Yu, F., et al. 2009. Talanta. 78: 1395-400. PMID: 19362207
- Ab initio study on the formation of triiodide CT complex from the reaction of iodine with 2,3-diaminopyridine. | Al-Hashimi, NA. and Hussein, YH. 2010. Spectrochim Acta A Mol Biomol Spectrosc. 75: 198-202. PMID: 19931482
- Spectroscopic characterization of charge transfer complexes of 2,3-diaminopyridine with chloranilic acid and dihydroxy-p-benzoquinone in polar solvent. | Al-Ahmary, KM. 2014. Spectrochim Acta A Mol Biomol Spectrosc. 117: 635-44. PMID: 24113016
- Charge transfer complex between 2,3-diaminopyridine with chloranilic acid. Synthesis, characterization and DFT, TD-DFT computational studies. | Al-Ahmary, KM., et al. 2018. Spectrochim Acta A Mol Biomol Spectrosc. 196: 247-255. PMID: 29454253
- Synthesis and evaluation of novel GSK-3β inhibitors as multifunctional agents against Alzheimer's disease. | Shi, XL., et al. 2019. Eur J Med Chem. 167: 211-225. PMID: 30772605
- Orange red-emitting carbon dots for enhanced colorimetric detection of Fe3. | Pu, J., et al. 2021. Analyst. 146: 1032-1039. PMID: 33300891
- Orange emissive carbon dots for fluorescent determination of hypoxanthine in fish. | Mou, Z., et al. 2022. Spectrochim Acta A Mol Biomol Spectrosc. 269: 120734. PMID: 34922290
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3-Diaminopyridine, 1 g | sc-254309 | 1 g | $29.00 | |||
2,3-Diaminopyridine, 5 g | sc-254309A | 5 g | $110.00 |
