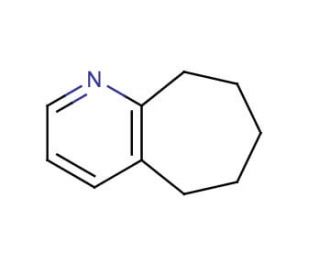
Molecular structure of 2,3-Cycloheptenopyridine, CAS Number: 7197-96-8
2,3-Cycloheptenopyridine (CAS 7197-96-8)
Alternate Names:
6,7,8,9-Tetrahydro-5H-cyclohepta[b]pyridine
CAS Number:
7197-96-8
Molecular Weight:
147.22
Molecular Formula:
C10H13N
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,3-Cycloheptenopyridine is a chemical compound that functions as a reactive intermediate in organic synthesis. It acts as a dienophile in Diels-Alder reactions, participating in cycloaddition reactions with dienes to form complex ring structures.2,3-Cycloheptenopyridine′s mechanism of action involves its ability to undergo cycloaddition reactions with electron-rich dienes, leading to the formation of fused ring systems with high regio- and stereo-selectivity. Through its reactivity as a dienophile, 2,3-Cycloheptenopyridine may play a role in the construction of diverse molecular architectures, contributing to the development of novel organic compounds in and development applications.
2,3-Cycloheptenopyridine (CAS 7197-96-8) References
- Indolizinones as synthetic scaffolds: fundamental reactivity and the relay of stereochemical information. | Hardin Narayan, AR. and Sarpong, R. 2012. Org Biomol Chem. 10: 70-8. PMID: 22072189
- Chichibabin-type direct alkylation of pyridyl alcohols with alkyl lithium reagents. | Jeffrey, JL. and Sarpong, R. 2012. Org Lett. 14: 5400-3. PMID: 23061536
- Synthesis and antiinflammatory activity of certain 5,6,7,8-tetrahydroquinolines and related compounds. | Calhoun, W., et al. 1995. J Med Chem. 38: 1473-81. PMID: 7739006
- Correlation of nonadditive kinetic effects with MINDO/3-derived molecular geometries | Jeffrey I. Seeman, Roseanne Galzerano, Keith Curtis, John C. Schug, and Jimmy W. Viers and Journal of the American Chemical Society. 1981. Journal of the American Chemical Society. 103 (19): 5982-5984.
- MINDO/3-based transition-state models for the menshutkin reaction. Iodomethylation of alkylpyridines | Jimmy W. Viers, John C. Schug, and Jeffrey I. Seeman. 1982. Journal of the American Chemical Society. 104 (3): 850-851.
- Thermolysis of Oxime O-Allyl Ethers: A New Method for Pyridine Synthesis | JUNKO KOYAMA; TERUYO SUGITA; YUKIO SUZUTA; HIROSHI IRIE. 1983. CHEMICAL & PHARMACEUTICAL. 31: 2601-2606.
- Correlation of nonadditive kinetic effects with molecular geometries. Structure and reactivity of alkyl- and cycloalkenylpyridines | Jeffrey I. Seeman, Jimmy W. Viers, John C. Schug, and Michael D. Stovall. 1984. Journal of the American Chemical Society. 106 (1): 143-151.
- Polyaza cavity-shaped molecules. Annelated derivatives of 2-(2'-pyridyl)-1,8-naphthyridine and 2,2'-bi-1,8-naphthyridine | Randolph P. Thummel, Francois Lefoulon, David Cantu, and Ramanathan Mahadevan. 1984. The Journal of Organic Chemistry. 49 (12): 2208-2212.
- Impact of phloretin and phloridzin on the formation of Maillard reaction products in aqueous models composed of glucose and L-lysine or its derivatives | Jinyu Ma; Xiaofang Peng; Kwan-Ming Ng; Chi-Ming Che; Mingfu Wang. 2012. Food Funct. 3: 178-186.
- Synthesis of 6, 7-dihydro-5 H-cyclopenta [b] pyridin-5-one analogues through manganese-catalyzed oxidation of the CH 2 adjacent to pyridine moiety in water | Lanhui Ren, Lianyue Wang, Ying Lv, Sensen Shang, Bo Chena, and and Shuang Gao. 2015. Green Chemistry. 17(4): 2369-2372.
- Optimization of Supercritical Extraction of Coriander (Coriandrum sativum L.) Seed and Characterization of Essential Ingredients | B.S. Shrirame, S.R. Geed, A. Raj, S. Prasad, M.K. Rai, A.K. Singh, R.S. Singh& B.N. Rai. 2018. Journal of Essential Oil Bearing Plants. 21,2018: 330-344.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3-Cycloheptenopyridine, 1 g | sc-230793 | 1 g | $54.00 |
