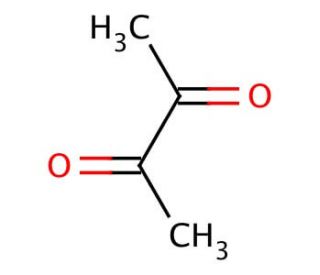
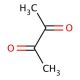
2,3-Butanedione (CAS 431-03-8)
QUICK LINKS
2,3-Butanedione, also known as Diacetyl, is a natural volatile organic compound (VOC) that occurs in various food items, beverages, and other consumer goods. It is formed as a byproduct during fermentation and possesses a distinctive buttery flavor and aroma. While 2,3-Butanedione has been extensively utilized in the food industry. The scientific community has conducted thorough investigations into 2,3-Butanedione, revealing its broad range of applications. It has served as a flavor enhancer in the food industry, a preservative in cosmetics. Inside the human body, 2,3-Butanedione undergoes metabolic processes facilitated by enzymes such as diacetyl reductase and diacetyl oxidase. These enzymes convert 2,3-Butanedione into acetoin, which is further metabolized into acetate and water. 2,3-Butanedione can also be transformed into other compounds like acetaldehyde and acetic acid.
2,3-Butanedione (CAS 431-03-8) References
- Origin of 2,3-pentanedione and 2,3-butanedione in D-glucose/L-alanine Maillard model systems. | Yaylayan, VA. and Keyhani, A. 1999. J Agric Food Chem. 47: 3280-4. PMID: 10552645
- Cyclocondensation of 2,3-butanedione in the presence of amino acids and formation of 4,5-dimethyl-1,2-phenylendiamine. | Guerra, PV. and Yaylayan, VA. 2013. Food Chem. 141: 4391-6. PMID: 23993630
- Breath gas metabolites and bacterial metagenomes from cystic fibrosis airways indicate active pH neutral 2,3-butanedione fermentation. | Whiteson, KL., et al. 2014. ISME J. 8: 1247-58. PMID: 24401860
- Chemical Reactivity and Respiratory Toxicity of the α-Diketone Flavoring Agents: 2,3-Butanedione, 2,3-Pentanedione, and 2,3-Hexanedione. | Morgan, DL., et al. 2016. Toxicol Pathol. 44: 763-83. PMID: 27025954
- EGFR-Dependent IL8 Production by Airway Epithelial Cells After Exposure to the Food Flavoring Chemical 2,3-Butanedione. | Kelly, FL., et al. 2019. Toxicol Sci. 169: 534-542. PMID: 30851105
- Generation of α-Diketones and 4-Hydroxy-2,5-dimethyl-3(2H)-furanone upon Coffee Roasting-Impact of Roast Degree on Reaction Pathways. | Poisson, L., et al. 2019. J Agric Food Chem. 67: 13829-13839. PMID: 30969764
- A potential flavor culture: Lactobacillus harbinensis M1 improves the organoleptic quality of fermented soymilk by high production of 2,3-butanedione and acetoin. | Zheng, Y., et al. 2020. Food Microbiol. 91: 103540. PMID: 32539956
- Toxicology and carcinogenesis studies of 2,3-butanedione in Wistar Han [Crl:WI(Han)] rats and B6C3F1/N mice (inhalation studies). | ,. 2018. Natl Toxicol Program Tech Rep Ser.. PMID: 33562897
- Molecular mechanism of two functional protein structure changes under 2,3-butanedione-induced oxidative stress and apoptosis effects in the hepatocytes. | Ni, X., et al. 2022. Int J Biol Macromol. 218: 969-980. PMID: 35907461
- Competitive Formation of 2,3-Butanedione and Pyrazines through Intervention of Added Cysteine during Thermal Processing of Alanine-Xylose Amadori Compounds. | Zhou, T., et al. 2022. J Agric Food Chem. 70: 15202-15212. PMID: 36444759
- Fungal pathogens causing postharvest fruit rot of wolfberry and inhibitory effect of 2,3-butanedione. | Ling, L., et al. 2022. Front Microbiol. 13: 1068144. PMID: 36704548
- 2,3-butanedione desensitization of bovine adrenal 2-oxoglutarate dehydrogenase to adenosine-5'-diphosphate. | Strumilo, SA., et al. 1984. Biomed Biochim Acta. 43: 237-40. PMID: 6732759
- Inactivation of L-lactate monooxygenase with 2,3-butanedione and phenylglyoxal. | Peters, RG., et al. 1981. Biochemistry. 20: 2564-71. PMID: 7236621
- 2,3-Butanedione inactivates the [3H]nitrendipine binding sites, whereas diethylpyrocarbonate does not. | Costa, B., et al. 1996. Neurochem Int. 29: 623-7. PMID: 9113129
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3-Butanedione, 1 ml | sc-238264 | 1 ml | $35.00 | |||
2,3-Butanedione, 5 ml | sc-238264A | 5 ml | $122.00 |
