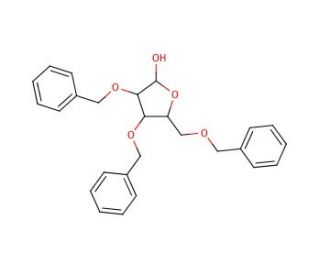
Molecular structure of 2,3,5-Tri-O-benzyl-β-D-arabinofuranose, CAS Number: 60933-68-8
2,3,5-Tri-O-benzyl-β-D-arabinofuranose (CAS 60933-68-8)
Application:
2,3,5-Tri-O-benzyl-β-D-arabinofuranose is an arabinofuranose derivative
CAS Number:
60933-68-8
Molecular Weight:
420.5
Molecular Formula:
C26H28O5
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,3,5-Tri-O-benzyl-β-D-arabinofuranose is a stereoselective analog that inhibits the human maltase glucoamylase and acetylation processes. It also has potent nucleophilic properties that allow it to react with the hydroxyl group of dimethyl fumarate, resulting in the formation of an acetal linkage. This compound is used in the stereo selective synthesis of oligosaccharides and carbohydrates.
2,3,5-Tri-O-benzyl-β-D-arabinofuranose (CAS 60933-68-8) References
- Palladium-catalyzed oxidation of benzylated aldose hemiacetals to lactones. | Bessmertnykh, A., et al. 2004. Carbohydr Res. 339: 1377-80. PMID: 15113678
- Synthesis of C8 alkyl glycosides via palladium-catalyzed telomerization of butadiene with O-benzylated aldoses. | Bessmertnykh, A., et al. 2006. Carbohydr Res. 341: 153-9. PMID: 16297889
- Spirocyclopropyl pyrrolidines as a new series of alpha-L-fucosidase inhibitors. | Laroche, C., et al. 2006. Bioorg Med Chem. 14: 4047-54. PMID: 16488612
- Synthesis of the C1-phosphonate analog of UDP-GlcNAc. | Chang, R., et al. 2006. Carbohydr Res. 341: 1998-2004. PMID: 16750179
- Synthesis of carbohydrate-based vinyl selenides via Wittig-type reactions. | Boutureira, O., et al. 2007. Carbohydr Res. 342: 736-43. PMID: 17258697
- Parallel synthesis of natural product-like polyhydroxylated pyrrolidine and piperidine alkaloids. | Chang, YF., et al. 2011. Mol Divers. 15: 203-14. PMID: 20563842
- Inhibition of fructose bisphosphatase and stimulation of phosphofructokinase by a stable isosteric phosphonate analog of fructose 2,6-bisphosphate. | McClard, RW., et al. 1986. Arch Biochem Biophys. 245: 282-6. PMID: 3004359
- TBAF Effects 3,6-Anhydro Formation from 6-O-Tosyl Pyranosides. | Morrison, ZA. and Nitz, M. 2020. Org Lett. 22: 1453-1457. PMID: 31990566
- Synthesis and Structural Revision of Glyphaeaside C. | Byatt, BJ., et al. 2021. Org Lett. 23: 4029-4033. PMID: 33929196
- Sharpless Asymmetric Dihydroxylation: An Impressive Gadget for the Synthesis of Natural Products: A Review. | Mushtaq, A., et al. 2023. Molecules. 28: PMID: 36985698
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,5-Tri-O-benzyl-β-D-arabinofuranose, 5 g | sc-216272 | 5 g | $184.00 |
