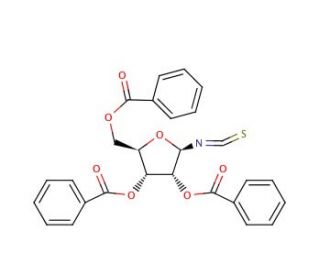
2,3,5-Tri-O-benzoyl-β-D-ribofuranosyl isothiocyanate (CAS 58214-53-2)
QUICK LINKS
2,3,5-Tri-O-benzoyl-β-D-ribofuranosyl isothiocyanate, a versatile chemical compound, has found significant utility in various research domains, particularly in organic synthesis and chemical biology. Its reactive isothiocyanate group facilitates conjugation reactions with amine-containing biomolecules, enabling the synthesis of labeled nucleosides, nucleotides, and peptides for biological studies. Researchers utilize its reactivity to selectively modify amino groups in proteins and peptides, facilitating the introduction of molecular probes or tags for protein labeling and imaging applications. Additionally, this compound serves as a valuable tool in the development of novel fluorescent probes and sensors for studying biological processes such as enzyme activity, protein-protein interactions, and cellular signaling pathways. Its ability to functionalize biomolecules with precision has made it an indispensable reagent in chemical biology research, enabling the elucidation of complex biological mechanisms at the molecular level. Furthermore, 2,3,5-Tri-O-benzoyl-β-D-ribofuranosyl isothiocyanate has been employed in the synthesis of nucleic acid analogs and modified oligonucleotides with potential applications in nucleic acid-based technologies, including diagnostics and nanotechnology. Overall, this compound′s unique reactivity and versatility make it a valuable asset in advancing research efforts across various scientific disciplines.
2,3,5-Tri-O-benzoyl-β-D-ribofuranosyl isothiocyanate (CAS 58214-53-2) References
- Chiral thioxohydroimidazoles with two sugar moieties. N-, C-, and spiro-nucleosides. | Gasch, Consolación, et al. 2000. Tetrahedron: Asymmetry. 11.2: 435-452.
- Stereocontrolled synthesis of thiohydantoin spironucleosides from sugar spiroacetals. | Fuentes, José, et al. 2006. Tetrahedron. 62.1: 97-111.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,5-Tri-O-benzoyl-β-D-ribofuranosyl isothiocyanate, 1 g | sc-256301 | 1 g | $244.00 |
