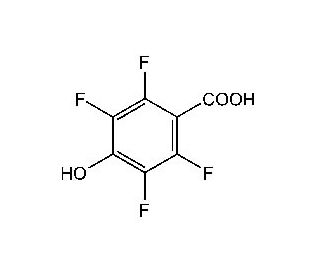
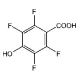
2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid (CAS 652-34-6)
QUICK LINKS
2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid is in materials science, particularly for its role in synthesizing advanced polymers and coatings with enhanced chemical resistance. Due to its fluorinated structure, 2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid hydrate contributes to the study of hydrophobic surfaces, which are useful in creating anti-corrosive and self-cleaning materials. In the field of organic chemistry, 2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid is used to investigate the mechanisms of aromatic substitution reactions, aiding in the understanding of electrophilic and nucleophilic processes on fluorinated aromatic rings. 2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid hydrate is explored in environmental chemistry for its potential in developing more stable and effective herbicides that are less prone to degradation by environmental factors.
2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid (CAS 652-34-6) References
- TEAC antioxidant activity of 4-hydroxybenzoates. | Tyrakowska, B., et al. 1999. Free Radic Biol Med. 27: 1427-36. PMID: 10641737
- Synthesis of certain 2'-deoxyuridine derivatives containing substituted phenoxy groups attached to C-5'; evaluation as potential dUTP analogues. | Marriott, JH., et al. 2001. Nucleosides Nucleotides Nucleic Acids. 20: 1691-704. PMID: 11580195
- Parallel synthesis and dopamine D3/D2 receptor screening of novel {4-[4-(2-methoxyphenyl)piperazin-1-yl]butyl}carboxamides. | Heidler, P., et al. 2005. Bioorg Med Chem. 13: 2009-14. PMID: 15727854
- Synthesis and antiplasmodial activity of new N-[3-(4-{3-[(7-chloroquinolin-4-yl)amino]propyl}piperazin-1-yl)propyl]carboxamides. | Freitag, M., et al. 2007. Bioorg Med Chem. 15: 2782-8. PMID: 17280835
- Synthesis of a series of N6-substituted adenosines with activity against trypanosomatid parasites. | Link, A., et al. 2009. Eur J Med Chem. 44: 3665-71. PMID: 19285758
- Recent developments in the design and synthesis of hybrid molecules based on aminoquinoline ring and their antiplasmodial evaluation. | Kouznetsov, VV. and Gómez-Barrio, A. 2009. Eur J Med Chem. 44: 3091-113. PMID: 19361896
- Improved quenched fluorescent probe for imaging of cysteine cathepsin activity. | Verdoes, M., et al. 2013. J Am Chem Soc. 135: 14726-30. PMID: 23971698
- Live Cell Imaging and Profiling of Cysteine Cathepsin Activity Using a Quenched Activity-Based Probe. | Edgington-Mitchell, LE., et al. 2017. Methods Mol Biol. 1491: 145-159. PMID: 27778287
- Manipulating Light-Induced Dynamic Macro-Movement and Static Photonic Properties within 1D Isostructural Hydrogen-Bonded Molecular Cocrystals. | Li, S., et al. 2020. Angew Chem Int Ed Engl. 59: 22623-22630. PMID: 32875702
- Mechanical Motion in Crystals Triggered by Solid State Photochemical [2+2] Cycloaddition Reaction. | Khan, S., et al. 2021. Chem Asian J. 16: 2806-2816. PMID: 34355513
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,5,6-Tetrafluoro-4-hydroxybenzoic acid, 5 g | sc-225637 | 5 g | $148.00 |
