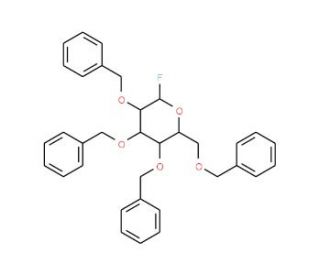
2,3,4,6-Tetra-O-benzyl-D-glucopyranosyl Fluoride (CAS 122741-44-0)
QUICK LINKS
2,3,4,6-Tetra-O-benzyl-D-glucopyranosyl fluoride is a highly effective glycosyl donor extensively used in glycoscience research, primarily for synthesizing complex carbohydrate structures. The benzyl groups protect the hydroxyl functionalities, enabling specific glycosylation reactions, while the fluoride moiety serves as an efficient leaving group. This structure allows for controlled stereoselectivity, favoring beta-glycosidic bond formation due to neighboring group participation. Researchers rely on this compound for constructing complex glycoconjugates, especially in the synthesis of oligosaccharides that mimic natural glycan structures. Its predictable reactivity and stable nature make it suitable for solid-phase and solution-phase glycosylation processes. Scientists exploit these properties to develop structurally precise glycomimetics, which are essential tools for understanding carbohydrate-binding interactions. The donor′s versatility allows for the formation of diverse glycosidic linkages that are crucial in exploring carbohydrate-protein interactions in molecular recognition and signal transduction. Furthermore, it facilitates the creation of glycan arrays and other high-throughput screening platforms to investigate protein-carbohydrate interactions. This is critical for deciphering the mechanisms of pathogen recognition, cell-cell communication, and immune responses. The controlled synthesis enabled by 2,3,4,6-Tetra-O-benzyl-D-glucopyranosyl fluoride provides researchers with valuable insights into the roles of carbohydrates in various biological systems.
2,3,4,6-Tetra-O-benzyl-D-glucopyranosyl Fluoride (CAS 122741-44-0) References
- Methods of synthesis of glycosyl fluorides. | Yokoyama, M. 2000. Carbohydr Res. 327: 5-14. PMID: 10968673
- Concise and scalable synthesis of aspalathin, a powerful plasma sugar-lowering natural product. | Han, Z., et al. 2014. J Nat Prod. 77: 583-8. PMID: 24354397
- Accelerated inverse electron demand Diels-Alder reactions of 1-oxa-1, 3-butadienes:[4+ 2] cycloaddition reactions of. beta.,. gamma.-unsaturated. alpha.-keto esters | Boger, D. L., & Robarge, K. D. 1988. The Journal of Organic Chemistry. 53(14): 3373-3377.
- A new method for the synthesis of fluoro-carbohydrates and glycosides using selectfluor | Burkart, M. D., Zhang, Z., Hung, S. C., & Wong, C. H. 1997. Journal of the American Chemical Society. 119(49): 11743-11746.
- Nucleophilic addition of 2-(trimethylsilyloxy) furan to a d-glucopyranose oxocarbenium ion | Jégou, A., & Veyrières, A. 1998. Tetrahedron: Asymmetry. 9(17): 3129-3134.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,4,6-Tetra-O-benzyl-D-glucopyranosyl Fluoride, 500 mg | sc-288402 | 500 mg | $309.00 |
