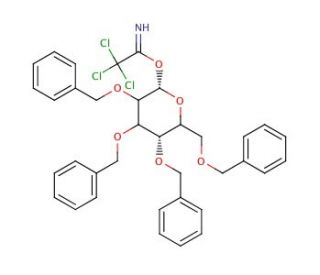
2,3,4,6-Tetra-O-benzyl-D-galactopyranose Trichloroacetimidate
QUICK LINKS
2,3,4,6-Tetra-O-benzyl-D-galactopyranose Trichloroacetimidate, a compound commonly abbreviated as ′TBDG-Trichloroacetimidate,′ is a valuable reagent widely employed in carbohydrate chemistry research. Its mechanism of action primarily involves its utility as a glycosyl donor for stereoselective glycosylation reactions. Upon activation with suitable promoters, TBDG-Trichloroacetimidate reacts with acceptor molecules, facilitating the formation of glycosidic bonds and allowing for the synthesis of complex glycoconjugates and oligosaccharides. This chemical modification is crucial for the efficient assembly of glycan structures with precise regio- and stereochemistry, enabling researchers to explore the diverse roles of carbohydrates in biological processes. TBDG-Trichloroacetimidate has been extensively used in the synthesis of biologically relevant glycoconjugates, glycopeptides, and glycolipids for studying carbohydrate-protein interactions, cell-cell recognition, and signaling pathways. Additionally, it finds application in the development of carbohydrate-based materials, such as glycan microarrays and glycodendrimers, for various research applications, including drug discovery, vaccine development, and diagnostics. Overall, TBDG-Trichloroacetimidate serves as a versatile tool in carbohydrate chemistry research, facilitating the synthesis of complex glycans and advancing our understanding of their roles in biology and medicine.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,4,6-Tetra-O-benzyl-D-galactopyranose Trichloroacetimidate, 250 mg | sc-213987 | 250 mg | $380.00 |
