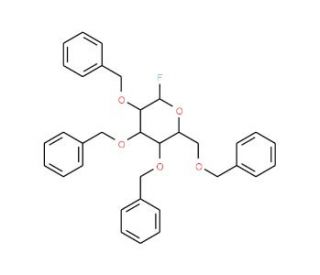
2,3,4,6-Tetra-O-benzyl-α-D-glucopyranosyl Fluoride (CAS 89025-46-7)
QUICK LINKS
2,3,4,6-Tetra-O-benzyl-α-D-glucopyranosyl fluoride is a chemical compound utilized primarily in carbohydrate chemistry research due to its ability to selectively block specific hydroxyl groups in sugar molecules. This selective protection strategy allows researchers to control the regioselective introduction of functional groups or modifications to specific positions within the sugar scaffold, enabling the synthesis of complex carbohydrate derivatives with tailored properties. In glycosylation reactions, the fluoride group serves as a temporary protecting group for the hydroxyl moiety at the anomeric carbon, preventing undesired reactions at this position. This compound has been extensively employed in the synthesis of oligosaccharides, glycoconjugates, and glycomimetics, facilitating the study of carbohydrate-protein interactions, cell surface recognition processes, and carbohydrate-based vaccine development. Moreover, the versatility of 2,3,4,6-Tetra-O-benzyl-α-D-glucopyranosyl fluoride in carbohydrate synthesis has led to its application in the preparation of glycosidase inhibitors, glycosyl transferase substrates, and carbohydrate-based materials with potential applications in biotechnology and materials science. Overall, this compound serves as a valuable tool for elucidating the roles of carbohydrates in various biological processes and for the development of carbohydrate-based probes.
2,3,4,6-Tetra-O-benzyl-α-D-glucopyranosyl Fluoride (CAS 89025-46-7) References
- An effective synthesis of isoorientin: the regioselective synthesis of a 6-C-glucosylflavone. | Kumazawa, T., et al. 2000. Carbohydr Res. 329: 507-13. PMID: 11128580
- Synthesis of Glycosyl Cyanides and C-Allyl Glycosides by the use of Glycosyl Fluoride Derivatives | Araki, Y., Kobayashi, N., Watanabe, K., & Ishido, Y. 1985. Journal of Carbohydrate Chemistry. 4(4): 565-585.
- Synthesis and enzyme-catalyzed hydrolysis of a radical-masked glycosylated spin-label reagent. | Sato, S., et al. 2004. Carbohydr Res. 339: 2425-32. PMID: 15388357
- Conversion of β-dC-glucopyranosyl phloroacetophenone to a spiroketal compound | Kumazawa, T., Asahi, N., Matsuba, S., Sato, S., Furuhata, K., & Onodera, J. I. 1998. Carbohydrate research. 308(1-2): 213-216.
- Regioselective acetyl transfer from the aglycon to the sugar in C-glycosylic compounds facilitated by silica gel | Kumazawa, T., Akutsu, Y., Matsuba, S., Sato, S., & Onodera, J. I. 1999. Carbohydrate research. 320(1-2): 129-137.
- Glycosyl fluorides from n-pentenyl-related glycosyl donors-Application to glycosylation strategies | Fraser-Reid, B., Lopez, J. C., Bernal-Albert, P., Gomez, A. M., Uriel, C., & Ventura, J. 2013. Canadian Journal of Chemistry. 91(1): 51-65.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,4,6-Tetra-O-benzyl-α-D-glucopyranosyl Fluoride, 500 mg | sc-288398 | 500 mg | $321.00 |
