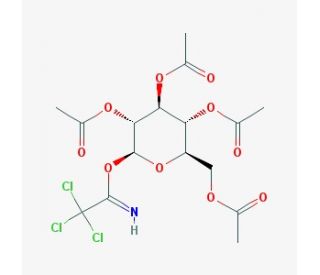
2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl 2,2,2-Trichloroacetimidate (CAS 92052-29-4)
QUICK LINKS
2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl 2,2,2-trichloroacetimidate is a valuable reagent in carbohydrate chemistry. Its structural features include the acetyl-protected hydroxyl groups that confer stability during chemical reactions and a trichloroacetimidate group, which acts as a highly efficient leaving group in glycosylation reactions. This compound is essential for glycosyl donor synthesis, enabling the attachment of various glycosyl residues to specific acceptor molecules. Researchers commonly use it in the synthesis of oligosaccharides, as it provides a reliable and controlled method for forming glycosidic linkages. In research, this compound is widely applied in the automated synthesis of complex oligosaccharides and glycoconjugates due to its predictable reactivity. It allows for the precise assembly of carbohydrate chains and facilitates regioselective glycosylation. Moreover, it has been used in the preparation of synthetic vaccines, glycomimetics, and glycan-based probes to study protein-glycan interactions and explain the structural biology of carbohydrates. The high reactivity of its trichloroacetimidate leaving group ensures high yields of glycosylation products, making it indispensable for the modular synthesis of complex glycan structures. This compound has thus enabled significant advancements in the study of carbohydrate-mediated molecular recognition, immune system research, and pathogen-host interactions.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl 2,2,2-Trichloroacetimidate, 1 g | sc-288394 | 1 g | $273.00 | |||
2,3,4,6-Tetra-O-acetyl-beta-D-glucopyranosyl 2,2,2-Trichloroacetimidate, 5 g | sc-288394A | 5 g | $950.00 |
