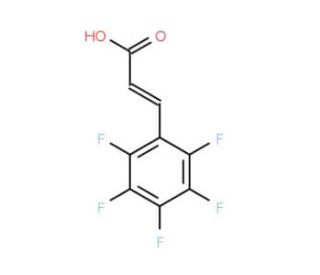
2,3,4,5,6-Pentafluorocinnamic acid (CAS 719-60-8)
QUICK LINKS
2,3,4,5,6-Pentafluorocinnamic acid (PFPA) is a versatile fluorinated molecule extensively utilized in scientific applications spanning organic synthesis, analytical chemistry, and biochemistry. Derived from cinnamic acid, an aromatic carboxylic acid, 2,3,4,5,6-Pentafluorocinnamic acid presents as a white, crystalline solid. 2,3,4,5,6-Pentafluorocinnamic acid finds extensive usage in various scientific research applications. In organic synthesis, it serves as a reagent for synthesizing diverse organic compounds. Additionally, it plays a role in analytical chemistry by enabling the detection and quantification of various organic compounds. In the field of biochemistry, 2,3,4,5,6-Pentafluorocinnamic acid acts as a substrate in enzyme assays and as a ligand in protein-ligand binding assays. Regarding its interaction with enzymes, 2,3,4,5,6-Pentafluorocinnamic acid serves as a substrate for a range of enzymes including esterases, amidases, and lipases. Furthermore, it acts as a ligand for proteins such as receptors and enzymes, forming interactions through hydrogen bonding and hydrophobic interactions. The binding of 2,3,4,5,6-Pentafluorocinnamic acid to these proteins and enzymes can influence their activity and function, making it a valuable compound for understanding their mechanisms.
2,3,4,5,6-Pentafluorocinnamic acid (CAS 719-60-8) References
- Characterisation of organometallic and coordination compounds by solvent-free matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry. | Wyatt, MF., et al. 2008. Analyst. 133: 47-8. PMID: 18087611
- Structural modification of the highly potent peptide bradykinin B1 receptor antagonist B9958. | Gera, L., et al. 2008. Int Immunopharmacol. 8: 289-92. PMID: 18182242
- Mapping the structural requirements of inducers and substrates for decarboxylation of weak acid preservatives by the food spoilage mould Aspergillus niger. | Stratford, M., et al. 2012. Int J Food Microbiol. 157: 375-83. PMID: 22726726
- Matrix-assisted laser desorption/ionization sample preparation optimization for structural characterization of poly(styrene-co-pentafluorostyrene) copolymers. | Tisdale, E., et al. 2014. Anal Chim Acta. 808: 151-62. PMID: 24370101
- Glow flow ionization mass spectrometry of small molecules. A comparison of a glow flow ionization source ('GlowFlow') with electrospray ionization and atmospheric pressure chemical ionization. | Owen, RN., et al. 2022. Rapid Commun Mass Spectrom. 36: e9327. PMID: 35610187
- Synthesis and binding of new polyfluorinated aryl azides to alpha-chymotrypsin. New reagents for photoaffinity labeling. | Soundararajan, N., et al. 1993. Bioconjug Chem. 4: 256-61. PMID: 8218481
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,4,5,6-Pentafluorocinnamic acid, 10 g | sc-280296 | 10 g | $459.00 |
