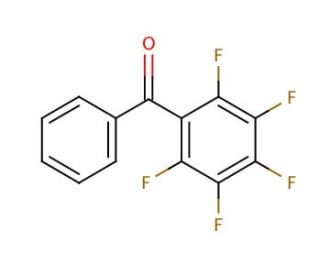
2,3,4,5,6-Pentafluorobenzophenone (CAS 1536-23-8)
QUICK LINKS
2,3,4,5,6-Pentafluorobenzophenone, referred to as PFB, is a synthetic organic compound characterized by its molecular formula, C13H4F5O. It presents as a colorless solid that readily dissolves in organic solvents and exhibits an exceptionally low vapor pressure. Owing to its distinctive properties, 2,3,4,5,6-Pentafluorobenzophenone has found widespread applications across various fields, including organic synthesis and materials science. Within the domain of organic synthesis, 2,3,4,5,6-Pentafluorobenzophenone serves as a valuable reagent, enabling the creation of diverse organic compounds like drugs, dyes, and polymers. Additionally, 2,3,4,5,6-Pentafluorobenzophenone plays a role in materials science, contributing to the synthesis of essential polymers like polyimides and polyurethanes. Although the precise mechanism of action for 2,3,4,5,6-Pentafluorobenzophenone remains incompletely understood, it is believed to function as an electron-deficient reagent, facilitating the formation of covalent bonds with electron-rich species. Moreover, 2,3,4,5,6-Pentafluorobenzophenone can act as an electrophile, engaging with nucleophiles to establish covalent bonds. This unique combination of characteristics makes 2,3,4,5,6-Pentafluorobenzophenone a versatile compound with significant importance in multiple scientific disciplines.
2,3,4,5,6-Pentafluorobenzophenone (CAS 1536-23-8) References
- Biological transformation, kinetics and dose-response assessments of bound musk ketone hemoglobin adducts in rainbow trout as biomarkers of environmental exposure. | Mottaleb, MA., et al. 2008. J Environ Sci (China). 20: 878-84. PMID: 18814586
- First regioselective cyclometalation reactions of cobalt in arylketones: C-H versus C-F activation. | Camadanli, S., et al. 2008. Dalton Trans. 5701-4. PMID: 18941655
- Relationship between odour-active compounds and flavour perception in meat from lambs fed different diets. | Resconi, VC., et al. 2010. Meat Sci. 85: 700-6. PMID: 20416794
- Color, lipid oxidation, sensory quality, and aroma compounds of beef steaks displayed under different levels of oxygen in a modified atmosphere package. | Resconi, VC., et al. 2012. J Food Sci. 77: S10-8. PMID: 22182210
- Highly fluorinated aryl-substituted tris(indazolyl)borate thallium complexes: diverse regiochemistry at the B-N bond. | Ojo, WS., et al. 2012. Inorg Chem. 51: 2893-901. PMID: 22339248
- Gas chromatographic-olfactometric aroma profile and quantitative analysis of volatile carbonyls of grilled beef from different finishing feed systems. | Resconi, VC., et al. 2012. J Food Sci. 77: S240-6. PMID: 22591324
- Functionalization of fluorinated molecules by transition-metal-mediated C-F bond activation to access fluorinated building blocks. | Ahrens, T., et al. 2015. Chem Rev. 115: 931-72. PMID: 25347593
- Efficient Access to Chiral Benzhydrols via Asymmetric Transfer Hydrogenation of Unsymmetrical Benzophenones with Bifunctional Oxo-Tethered Ruthenium Catalysts. | Touge, T., et al. 2016. J Am Chem Soc. 138: 10084-7. PMID: 27463264
- Magnetic field effects on the behavior of radicals in protein and DNA environments. | Mohtat, N., et al. 1998. Photochem Photobiol. 67: 111-8. PMID: 9477768
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,3,4,5,6-Pentafluorobenzophenone, 5 g | sc-260217 | 5 g | $91.00 | |||
2,3,4,5,6-Pentafluorobenzophenone, 25 g | sc-260217A | 25 g | $234.00 |
