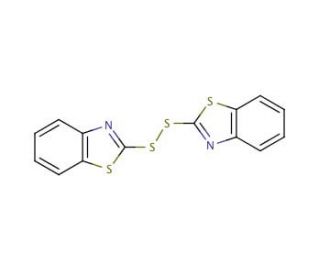
2,2′-Dithiobis(benzothiazole) (CAS 120-78-5)
QUICK LINKS
2,2′-Dithiobis(benzothiazole) is a chemical compound that functions as an accelerator in the vulcanization of rubber. It acts as a sulfur donor during the cross-linking process, promoting the formation of strong and durable rubber products. This compound works by reacting with the rubber polymer chains, leading to the formation of cross-links between the chains. These cross-links enhance the mechanical properties of the rubber, such as its strength, elasticity, and resistance to heat and aging. In experimental applications, 2,2′-Dithiobis(benzothiazole) may play a role in the modification of rubber properties, contributing to the development of high-performance rubber materials for various industrial applications. Its mode of action involves facilitating the cross-linking process, ultimately leading to the enhancement of rubber′s mechanical and thermal properties.
2,2′-Dithiobis(benzothiazole) (CAS 120-78-5) References
- Organosulfur compounds as pre-exposure therapy for threat agents. | Ternay, AL., et al. 2000. J Appl Toxicol. 20 Suppl 1: S31-4. PMID: 11428640
- Oxidation of 2-mercaptobenzothiazole in latex gloves and its possible haptenation pathway. | Chipinda, I., et al. 2007. Chem Res Toxicol. 20: 1084-92. PMID: 17630704
- Metal-Free Preparation of Cycloalkyl Aryl Sulfides via Di-tert-butyl Peroxide-Promoted Oxidative C(sp3)[BOND]H Bond Thiolation of Cycloalkanes. | Zhao, J., et al. 2014. Adv Synth Catal. 356: 2719-2724. PMID: 25505857
- Photochemical reaction dynamics of 2,2'-dithiobis(benzothiazole): direct observation of the addition product of an aromatic thiyl radical to an alkene with time-resolved vibrational and electronic absorption spectroscopy. | Koyama, D. and Orr-Ewing, AJ. 2016. Phys Chem Chem Phys. 18: 12115-27. PMID: 27076054
- Synthesis, properties, antitumor and antibacterial activity of new Pt(II) and Pd(II) complexes with 2,2'-dithiobis(benzothiazole) ligand. | Rubino, S., et al. 2017. Bioorg Med Chem. 25: 2378-2386. PMID: 28336408
- Synthesis and initial screening of lactate dehydrogenase inhibitor activity of 1,3-benzodioxole derivatives. | Annas, D., et al. 2020. Sci Rep. 10: 19889. PMID: 33199724
- Palladium nanoparticles immobilized on a nano-silica triazine dendritic polymer: a recyclable and sustainable nanoreactor for C-S cross-coupling. | Landarani-Isfahani, A., et al. 2020. RSC Adv. 10: 21198-21205. PMID: 35518753
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2′-Dithiobis(benzothiazole), 100 g | sc-238249 | 100 g | $50.00 |
