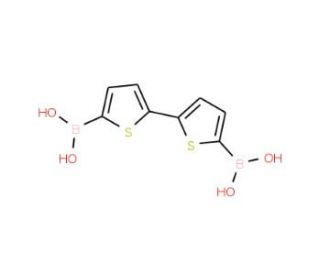
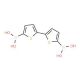
2,2′-Bithiophene-5,5′-diboronic acid (CAS 189358-30-3)
QUICK LINKS
2,2′-Bithiophene-5,5′-diboronic acid (BTDBA) is a boronic acid derivative of the heterocyclic compound bithiophene. Bithiophene compounds with boronic acid groups are often utilized in the synthesis of conjugated polymers and small organic molecules that have applications in organic electronics, such as organic photovoltaic cells, organic field-effect transistors, and organic light-emitting diodes. The boronic acid functionalities allow for Suzuki-Miyaura cross-coupling reactions, a powerful tool for forming carbon-carbon bonds in organic synthesis. 2,2′-Bithiophene-5,5′-diboronic acid acts via a variety of mechanisms, including inhibition of cell proliferation, induction of apoptosis, and inhibition of angiogenesis. 2,2′-Bithiophene-5,5′-diboronic acid may act to inhibit the growth of bacteria, fungi, viruses, and cancer cells. It also has anti-inflammatory, antioxidant, and anti-angiogenic properties.
2,2′-Bithiophene-5,5′-diboronic acid (CAS 189358-30-3) References
- Red-Shifts upon Metal Binding: A Di-Gold(I)-Substituted Bithiophene | Miya A. Peay, James E. Heckler, Nihal Deligonul, and Thomas G. Gray*. 2011,. Organometallics. 30, 18,: 5071–5074.
- Utility of a heterogeneous palladium catalyst for the synthesis of a molecular semiconductor via Stille, Suzuki, and direct heteroarylation cross-coupling reactions† | Seth M. McAfee, Jenny S. J. McCahill, Casper M. Macaulay, Arthur D. Hendsbee and Gregory C. Welch *. 2015,. RSC Adv.,. 5,: 26097-26106.
- High-performance semiconductors based on oligocarbazole–thiophene derivatives for solution-fabricated organic field-effect transistors | Gung-Pei Chang, Kuo-Huang Hsieh. 1 January 2013,. Thin Solid Films. Volume 527,: Pages 291-298.
- Thermoresponsive core–shell nanoparticles with cross-linked π-conjugate core based on amphiphilic block copolymers by RAFT polymerization and palladium … | Yohei Abiko b, Aiko Matsumura a, Kazuhiro Nakabayashi a, Hideharu Mori a b. 5 November 2014,. Polymer. Volume 55, Issue 23,: Pages 6025-6035.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2′-Bithiophene-5,5′-diboronic acid, 500 mg | sc-298559 | 500 mg | $163.00 | |||
2,2′-Bithiophene-5,5′-diboronic acid, 1 g | sc-298559A | 1 g | $271.00 |
