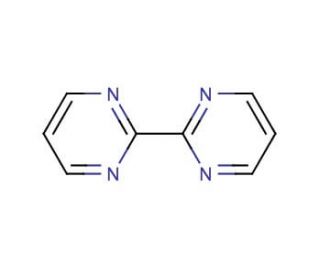
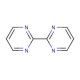
Molecular structure of 2,2′-Bipyrimidine, CAS Number: 34671-83-5
2,2′-Bipyrimidine (CAS 34671-83-5)
Alternate Names:
2,2′-Dipyrimidyl
CAS Number:
34671-83-5
Purity:
≥98%
Molecular Weight:
158.16
Molecular Formula:
C8H6N4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,2′-Bipyrimidine is a heterocyclic organic compound and a derivative of pyrimidine that is used in a variety of scientific research applications. It is used in the synthesis of polymers, dyes, pigments, and other heterocyclic compounds. It acts as a nucleophile in a condensation reaction and an electrophile in a substitution reaction. 2,2′-Bipyrimidine is known to be a relatively non-toxic compound and an inhibitor of the enzyme cytochrome P450.
2,2′-Bipyrimidine (CAS 34671-83-5) References
- 1,2,4,5-benzenetetracarboxylate- and 2,2'-bipyrimidine-containing cobalt(II) coordination polymers: preparation, crystal structure, and magnetic properties. | Fabelo, O., et al. 2008. Inorg Chem. 47: 3568-76. PMID: 18396860
- Monomeric Ti(iv)-based complexes incorporating luminescent nitrogen ligands: synthesis, structural characterization, emission spectroscopy and cytotoxic activities. | Khalil, G., et al. 2016. Dalton Trans. 45: 19072-19085. PMID: 27858023
- The Diphosphorus Complex [Cp2Mo2(CO)4(η2-P2)] as a Building Block for the Synthesis of Mixed-Hybrid Coordination Polymers. | Moussa, ME., et al. 2016. Eur J Inorg Chem. 2016: 4538-4541. PMID: 27867315
- High-Voltage Dye-Sensitized Solar Cells Mediated by [Co(2,2'-bipyrimidine)3]z. | Chen, KY., et al. 2017. Inorg Chem. 56: 2383-2386. PMID: 28218525
- Molecular Design of Thermally Activated Delayed-Fluorescent Emitters Using 2,2'-Bipyrimidine as the Acceptor in Donor-Acceptor Structures. | Park, HJ., et al. 2017. Chem Asian J. 12: 2494-2500. PMID: 28695621
- Visible Light-Activated CO Release and 1O2 Photosensitizer Formation with Ru(II),Mn(I) Complexes. | Pickens, RN., et al. 2018. Inorg Chem. 57: 11616-11625. PMID: 30160480
- Remarkable Changes of the Acidity of Bound Nitroxyl (HNO) in the [Ru(Me3[9]aneN3)(L2)(NO)] n+ Family (n = 1-3). Systematic Structural and Chemical Exploration and Bioinorganic Chemistry Implications. | Levin, N., et al. 2018. Inorg Chem. 57: 12270-12281. PMID: 30222334
- Substituent Effects on Exchange Coupling and Magnetic Relaxation in 2,2'-Bipyrimidine Radical-Bridged Dilanthanide Complexes. | Gould, CA., et al. 2020. J Am Chem Soc. 142: 21197-21209. PMID: 33322909
- Interrogation of 2,2'-Bipyrimidines as Low-Potential Two-Electron Electrolytes. | Griffin, JD., et al. 2021. J Am Chem Soc. 143: 992-1004. PMID: 33411535
- 2,2'-Bipyrimidine-based luminescent Ru(ii)/Ir(iii)-arene monometallic and homo- and hetero-bimetallic complexes for therapy against MDA-MB-468 and caco-2 cells. | Roy, N., et al. 2021. Dalton Trans. 50: 11725-11729. PMID: 34612310
- Ionic indium(III) chloride hybrids incorporating a 2,2'-bipyrimidine ligand: studies on photoluminescence and structural transformation. | Jin, JC., et al. 2021. Dalton Trans. 50: 16406-16413. PMID: 34734938
- Modified pyridine-triazole and 2,2'-bipyrimidine ligands generating robust titanium complexes constructed around a TiO4N2 core. | Barloy, L., et al. 2021. Dalton Trans. 50: 17008-17022. PMID: 34738604
- Electron Shuttle in N-Heteroaromatic Ni Catalysts for Alkene Isomerization. | Tricoire, M., et al. 2022. JACS Au. 2: 1881-1888. PMID: 36032537
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2′-Bipyrimidine, 1 g | sc-254304 | 1 g | $209.00 |
