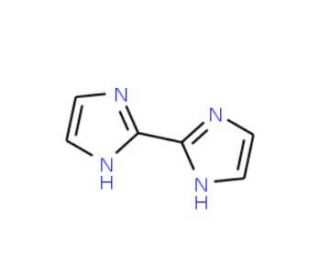
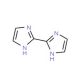
2,2′-Biimidazole (CAS 492-98-8)
QUICK LINKS
2,2′-Biimidazole is a white powder and demonstrates solubility in polar solvents like methanol, water, and dimethyl sulfoxide (DMSO). This compound acts as a Lewis base, capable of coordinating with metal ions such as Cu(I), Ag(I), and Au(I-II). The resultant coordination compounds have exhibited catalytic activity superior to other metal-ligand complexes. 2,2′-Biimidazole is used as a building block in the construction of supramolecular structures, nanoparticles, and other coordination compounds. Its role as a ligand in catalysis contributes to enhanced efficiency in chemical reactions. Additionally, 2,2′-Biimidazole has also been used in the synthesis of fluorescent proteins, facilitating the exploration of protein structure and function within cells.
2,2′-Biimidazole (CAS 492-98-8) References
- Bis[2-(1H-imidazol-2-yl-κN)-1H-imidazol-3-ium]silver(I) trinitrate. | Finch, SR., et al. 2011. Acta Crystallogr Sect E Struct Rep Online. 67: m909. PMID: 21836895
- A Comprehensive Study on the Dye Adsorption Behavior of Polyoxometalate-Complex Nano-Hybrids Containing Classic β-Octamolybdate and Biimidazole Units. | Liang, S., et al. 2019. Molecules. 24: PMID: 30813387
- Inhibition of autophagic flux by cyclometalated iridium(iii) complexes through anion transportation. | Chen, MH., et al. 2019. Chem Sci. 10: 3315-3323. PMID: 30996918
- Similarities and differences in d6 low-spin ruthenium, rhodium and iridium half-sandwich complexes: synthesis, structure, cytotoxicity and interaction with biological targets. | Gilewska, A., et al. 2019. J Biol Inorg Chem. 24: 591-606. PMID: 31115765
- Cross-Electrophile Coupling of Unactivated Alkyl Chlorides. | Sakai, HA., et al. 2020. J Am Chem Soc. 142: 11691-11697. PMID: 32564602
- Catalytic trifluoromethylation of iodoarenes by use of 2-trifluoromethylated benzimidazoline as trifluoromethylating reagent. | Uchikura, T., et al. 2020. Beilstein J Org Chem. 16: 2442-2447. PMID: 33082878
- Phosphorescent metal complexes as theranostic anticancer agents: combining imaging and therapy in a single molecule. | Tan, CP., et al. 2021. Chem Sci. 12: 2357-2367. PMID: 34164000
- Interaction of a 1,3-Dicarbonyl Toxin with Ru(II)-Biimidazole Complexes for Luminescence Sensing: A Spectroscopic and Photochemical Experimental Study Rationalized by Time-Dependent Density Functional Theory Calculations. | Quílez-Alburquerque, J., et al. 2022. Inorg Chem. 61: 328-337. PMID: 34923820
- Insights on the Synthesis of N-Heterocycles Containing Macrocycles and Their Complexion and Biological Properties. | Malek, F., et al. 2022. Molecules. 27: PMID: 35408522
- Unexpected molecular diversity of brown carbon formed by Maillard-like reactions in aqueous aerosols. | Tang, S., et al. 2022. Chem Sci. 13: 8401-8411. PMID: 35919720
- RUNAT-BI: A Ruthenium(III) Complex as a Selective Anti-Tumor Drug Candidate against Highly Aggressive Cancer Cell Lines. | Albanell-Fernández, M., et al. 2022. Cancers (Basel). 15: PMID: 36612065
- Bipyridine Ruthenium(II) Complexes with Halogen-Substituted Salicylates: Synthesis, Crystal Structure, and Biological Activity. | Schoeller, M., et al. 2023. Molecules. 28: PMID: 37375164
- Recent advances in fluorescent materials for mercury(ii) ion detection. | Li, Q. and Zhou, Y. 2023. RSC Adv. 13: 19429-19446. PMID: 37383685
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2′-Biimidazole, 1 g | sc-298557 | 1 g | $84.00 | |||
2,2′-Biimidazole, 5 g | sc-298557A | 5 g | $388.00 |
