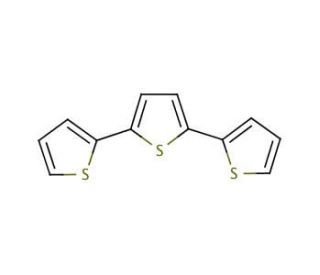
2,2′:5′,2′′-Terthiophene (CAS 1081-34-1)
QUICK LINKS
2,2′:5′,2′′-Terthiophene (2,2′:5′,2′′-TTP) is a member of the thiophene family of organic compounds. Its unique structure features a five-membered ring connecting two sulfur atoms and a two-membered ring connecting two oxygen atoms. Extensively studied for its remarkable characteristics, 2,2′:5′,2′′-Terthiophene exhibits high solubility in organic solvents, making it of great interest for potential applications in organic electronics and optoelectronics.In the realm of organic electronics and optoelectronics, 2,2′:5′,2′′-Terthiophene takes center stage. It serves as a active material in devices like organic field-effect transistors (OFETs), organic light-emitting diodes (OLEDs), and organic photovoltaic cells (OPVs). It plays a pivotal role in the development of sensors, particularly electrochemical sensors, capable of detecting diverse substances such as glucose, ammonia, and nitric oxide. One of its key attributes lies in its ability to act as an electron donor, facilitating strong interactions with other molecules, especially those encountered in the realms of organic electronics and optoelectronics.
2,2′:5′,2′′-Terthiophene (CAS 1081-34-1) References
- Photo-induced transformations in 2,2':5',2''-terthiophene thin films on silver. | Alaverdyan, Y., et al. 2006. Phys Chem Chem Phys. 8: 1445-50. PMID: 16633627
- elective and efficient syntheses of phototoxic 2, 2′: 5′, 2 ″-terthiophene derivatives bearing a functional substituent in the 3′-or the 5-position | Rossi, R., Carpita, A., Ciofalo, M., & Lippolis, V. 1991. Tetrahedron. 47(39): 8443-8460.
- Singlet-triplet intersystem crossing in 2, 2′: 5′, 2 ″-terthiophene and some of its derivatives. | Rossi, R., Ciofalo, M., Carpita, A., & Ponterini, G. 1993. Journal of Photochemistry and Photobiology A: Chemistry. 70(1): 59-67.
- Mechanistic Study of the Photooxidation of Squalene Sensitized with 2, 2′: 5′, 2 ″-Terthiophene and 2, 2′-Bithiophene | Fujita, H., Tokiwa, K., Sayama, K., Mori, H., & Sasaki, M. 1993. Bulletin of the Chemical Society of Japan. 66(10): 2960-2964.
- Oxidative addition reactions of 2, 2′-bithiophene and 2, 2′: 5′, 2 ″-terthiophene with trinuclear clusters of iron, ruthenium and osmium | Arce, A. J., Arrojo, P., De Sanctis, Y., Márquez, M., & Deeming, A. J. 1994. Journal of organometallic chemistry. 479(1-2): 159-164.
- A theoretical study of 2, 2′; 5′, 2 ″-terthiophene (α-T) and its analogs. Part 1. Correlation of electronic structure and energies with herbicidal phototoxicity | Friedman, D. C., & Friedman, P. 1995. Journal of Molecular Structure: THEOCHEM,. 333(1-2): 71-78.
- Theoretical investigations on positive bipolaronic defects in oligothiophenes: Structures and vibrational spectra of doubly charged 2,2′-bithiophene and 2,2′:5′,2″-terthiophene | Ehrendorfer, C., & Karpfen, A. 1995. Vibrational Spectroscopy. 8(2): 293-303.
- Stationary and time-resolved spectra of 2, 2′: 5′, 2 ″-terthiophene | Yang, A., Hughes, S., Kuroda, M., Shiraishi, Y., & Kobayashi, T. 1997. Chemical physics letters. 280(5-6): 475-480.
- Novel diphenylphosphine derivatives of 2,2′-bithiophene, 2,2′:5′,2″-terthiophene, 2-(2′-thienyl)pyridine and 2,6-di-2′-thienylpyridine. Crystal structures of 5,5′-bis(diphenylphosphino)-2,2′-bithiophene, diphenyl{5-[6′-(diphenylphosphino)-2′-pyridyl]-2-thienyl}phosphine and 2,6-bis[5′-(diphenylphosphino)-2′-thienyl]pyridine | Field, J. S., Haines, R. J., Lakoba, E. I., & Sosabowski, M. H. 2001. Journal of the Chemical Society, Perkin Transactions 1. (24): 3352-3360.
- Electropolymerization of 2,2′:5′,2″ terthiophene at an electrified liquid–liquid interface | Gorgy, K., Fusalba, F., Evans, U., Kontturi, K., & Cunnane, V. J. 2001. Synthetic metals. 125(3): 365-373.
- Spectroelectrochemical Study of 2,2':5',2' '-Terthiophene Polymerization at a Liquid/Liquid Interface Controlled by Potential-Determining Ions | Evans-Kennedy, U., Clohessy, J., & Cunnane, V. J. 2004. Macromolecules. 37(10): 3630-3634.
- Electrochemical copolymerization of carbazole and 2, 2′: 5′-2 ″terthiophene: characterization and micro-capacitor application | Ates, M., & Eren, N. 2014. Iranian Polymer Journal. 23: 581-589.
- Ionic liquid-assisted vapor deposition and polymorphs control of 2, 2′: 5′, 2 ″-terthiophene crystals | Okawara, K., Maruyama, S., & Matsumoto, Y. 2019. Japanese Journal of Applied Physics. 58(8): 085503.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2′:5′,2′′-Terthiophene, 1 g | sc-256272 | 1 g | $112.00 |
