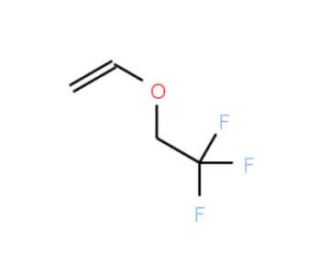
2,2,2-Trifluoroethyl vinyl ether (CAS 406-90-6)
QUICK LINKS
2,2,2-Trifluoroethyl vinyl ether is a substance that is used as a solvent in the manufacturing of various chemicals. In materials science and polymer chemistry, 2,2,2-Trifluoroethyl vinyl ether stands out for its potential as a monomer in the synthesis of fluorinated polymers. Polymers derived from this compound are expected to exhibit enhanced chemical resistance, thermal stability, and hydrophobicity due to the fluorinated side chain. This compound binds to cytochrome P450 enzymes, consequently inhibiting their activity. It is believed that this inhibitory effect arises from the generation of hydrogen fluoride (HF) or water vapor as byproducts during the reaction. Moreover, 2,2,2-Trifluoroethyl vinyl ether has the ability to react with halothane or nitrous oxide (N2O), leading to the formation of these substances. Additionally, it can react with oxygen (O) to produce ozone (O3).
2,2,2-Trifluoroethyl vinyl ether (CAS 406-90-6) References
- 19F nuclear magnetic resonance analysis of trifluoroethanol metabolites in the urine of the Sprague-Dawley rat. | Selinsky, BS., et al. 1991. Biochem Pharmacol. 42: 2229-38. PMID: 1958238
- Metabolism and toxicity of 2,2,2-trifluoroethyl vinyl ether. | Fiserova-Bergerova, V. 1977. Environ Health Perspect. 21: 225-30. PMID: 25763
- Potential energy surface of fluoroxene: experiment and theory. | Uriarte, I., et al. 2016. Phys Chem Chem Phys. 18: 3966-74. PMID: 26771032
- Potentiation of fluroxene (2,2,2-trifluoroethyl vinyl ether) toxicity with polychlorinated biphenyls. | Murphy, MJ., et al. 1979. Toxicol Appl Pharmacol. 48: 87-97. PMID: 36687
- Mutagenicity of inhalation anaesthetics: trichloroethylene, divinyl ether, nitrous oxide and cyclopropane. | Baden, JM., et al. 1979. Br J Anaesth. 51: 417-21. PMID: 375954
- Destruction of cytochrome P-450 by vinyl fluoride, fluroxene, and acetylene. Evidence for a radical intermediate in olefin oxidation. | Ortiz de Montellano, PR., et al. 1982. Biochemistry. 21: 1331-9. PMID: 6122467
- Reaction schemes for the degradation of cytochrome P-450 by allyl-iso-propylacetamide and fluroxene. | Ivanetich, KM., et al. 1981. Adv Exp Med Biol. 136 Pt B: 1213-20. PMID: 6124093
- Trifluorinated ether anesthetic lethality in rats: the role of bacterial infection. | Dersham, G., et al. 1983. Toxicol Appl Pharmacol. 71: 93-100. PMID: 6138884
- Absence of mutagenic activity of trifluoroethanol and its metabolites in Salmonella typhimurium. | Blake, DA., et al. 1981. Fundam Appl Toxicol. 1: 415-8. PMID: 6764424
- The role of cytochrome P-450 in the toxicity of fluroxene (2,2,2-trifluoroethyl vinyl ether) anaesthesia in vivo. | Ivanetich, KM., et al. 1976. Biochem Pharmacol. 25: 773-8. PMID: 7270
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,2,2-Trifluoroethyl vinyl ether, 100 mg | sc-230728 | 100 mg | $161.00 |
