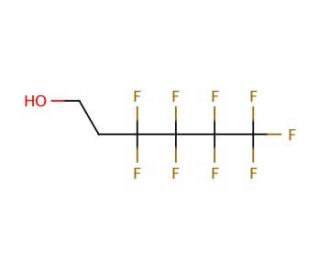
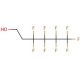
1H,1H,2H,2H-Perfluorohexan-1-ol (CAS 2043-47-2)
QUICK LINKS
1H,1H,2H,2H-Perfluorohexan-1-ol is a colorless liquid characterized by its low boiling point and low melting point. Although insoluble in water, it readily dissolves in organic solvents. This fluorinated organic compound has found widespread use in various scientific applications, encompassing synthesis, drug development, and environmental monitoring. Within the realm of scientific research, 1H,1H,2H,2H-Perfluorohexan-1-ol has played a significant role. It serves as a versatile solvent for synthesizing a range of organic compounds and as a valuable reagent in organic reactions. Additionally, it acts as a catalyst in the production of polymer materials. The compound has further contributed to pharmaceutical synthesis, aiding in the development of novel drugs. Furthermore, it has proved beneficial for environmental monitoring, particularly in assessing the quality of water and air. The fluorinated nature of 1H,1H,2H,2H-Perfluorohexan-1-ol imparts a high affinity for specific organic compounds. It exhibits interactions with hydrophobic regions of proteins, lipids, and other organic molecules, enabling its utility as a solvent, catalyst, or reagent in organic reactions. Moreover, its interactions with hydrophilic regions of proteins and lipids allow it to act as a surfactant, facilitating the absorption of other organic molecules. In addition to its practical applications, 1H,1H,2H,2H-Perfluorohexan-1-ol has displayed biochemical and physiological effects. It demonstrates weak inhibitory properties against certain enzymes, including cytochrome P450 enzymes and proteases. Additionally, it acts as a mild agonist of specific G-protein coupled receptors, such as muscarinic acetylcholine receptors. Furthermore, the compound exhibits antioxidant properties, contributing to its overall biological significance.
1H,1H,2H,2H-Perfluorohexan-1-ol (CAS 2043-47-2) References
- Fluorinated ionic surfactant microemulsions in hydrofluorocarbon 134a (HFC 134a). | Patel, N., et al. 2003. J Colloid Interface Sci. 258: 354-62. PMID: 12618105
- Comparison of three types of mass spectrometers for HPLC/MS analysis of perfluoroalkylated substances and fluorotelomer alcohols. | Berger, U., et al. 2004. Eur J Mass Spectrom (Chichester). 10: 579-88. PMID: 15531790
- Reverse water-in-fluorocarbon microemulsions stabilized by new polyhydroxylated nonionic fluorinated surfactants. | Debbabi, K., et al. 2009. Langmuir. 25: 8919-26. PMID: 19344159
- Negative-ion atmospheric pressure ionisation of semi-volatile fluorinated compounds for ultra-high-performance liquid chromatography tandem mass spectrometry analysis. | Ayala-Cabrera, JF., et al. 2018. Anal Bioanal Chem. 410: 4913-4924. PMID: 29796902
- A Micelle Electrolyte Enabled by Fluorinated Ether Additives for Polysulfide Suppression and Li Metal Stabilization in Li-S Battery. | Zhao, Y., et al. 2020. Front Chem. 8: 484. PMID: 32637395
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1H,1H,2H,2H-Perfluorohexan-1-ol, 25 g | sc-223204 | 25 g | $258.00 |
