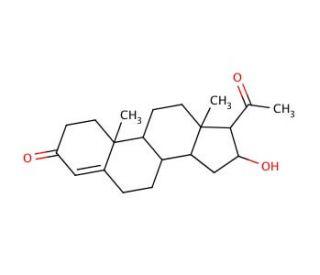
Molecular structure of 16α-Hydroxy Progesterone
16α-Hydroxy Progesterone (CAS 438-07-3)
Alternate Names:
16α-Hydroxy-4-pregnene-3,20-dione; 16α-Hydroxy-Δ4-pregnene-3,20-dione; 16α-Hydroxyprogesterone
Application:
16α-Hydroxy Progesterone is a human progesterone receptors hPR-A and hPR-B agonist
CAS Number:
438-07-3
Molecular Weight:
330.46
Molecular Formula:
C21H30O3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
16alpha-Hydroxy Progesterone a hydroxylated metabolite of Progesterone that binds to human progesterone receptors hPR-A and hPR-B and act as an agonist for both receptors. 16alpha-Hydroxy progesterone exerts its effects in the body through multiple mechanisms. It interacts with the progesterone receptor, present in various tissues, stimulating its activation. This, in turn, triggers a series of signaling pathways that modulate gene expression and promote the synthesis of diverse proteins and hormones. Moreover, 16alpha-Hydroxy progesterone can bind to and activate additional receptors, including the androgen receptor, potentially resulting in testosterone production.
16α-Hydroxy Progesterone (CAS 438-07-3) References
- 16alpha-hydroxy-progesterone as a sex hormone antagonist: effect on venous diameters in the female mouse. | Forbes, TR. and Taku, E. 1975. Anat Rec. 182: 67-70. PMID: 1171637
- IS THERE A POTENT NATURALLY OCCURRING SODIUM-LOSING STEROID HORMONE? | GEORGE, JM., et al. 1965. J Clin Endocrinol Metab. 25: 621-7. PMID: 14313756
- Differential action of progesterones on hepatic microsomal activities in the rat. | Feuer, G., et al. 1977. Biochem Pharmacol. 26: 1495-9. PMID: 197967
- Role of CYP2A5 in the clearance of nicotine and cotinine: insights from studies on a Cyp2a5-null mouse model. | Zhou, X., et al. 2010. J Pharmacol Exp Ther. 332: 578-87. PMID: 19923441
- Neuroactive steroids in periphery and cerebrospinal fluid. | Kancheva, R., et al. 2011. Neuroscience. 191: 22-7. PMID: 21641969
- Dydrogesterone metabolism in human liver by aldo-keto reductases and cytochrome P450 enzymes. | Olbrich, M., et al. 2016. Xenobiotica. 46: 868-74. PMID: 26796435
- Characterization of Maternal and Fetal CYP3A-Mediated Progesterone Metabolism. | Quinney, SK., et al. 2017. Fetal Pediatr Pathol. 36: 400-411. PMID: 28949811
- Progesterone Metabolites Inhibit the Human Ether-a-go-go-Related Gene and Predict QT Interval Length. | Shugg, T., et al. 2020. J Clin Pharmacol. 60: 648-659. PMID: 31829451
- Metabolic Profiling and Quantitative Analysis of Cerebrospinal Fluid Using Gas Chromatography-Mass Spectrometry: Current Methods and Future Perspectives. | Pautova, A., et al. 2021. Molecules. 26: PMID: 34208377
- Powerful Steroid-Based Chiral Selector for High-Throughput Enantiomeric Separation of α-Amino Acids Utilizing Ion Mobility-Mass Spectrometry. | Li, Y., et al. 2021. Anal Chem. 93: 13589-13596. PMID: 34597017
- Association of genetic polymorphisms with local steroid metabolism in human benign breasts. | Lee, O., et al. 2022. Steroids. 177: 108937. PMID: 34762930
- Metabolism of 16alpha-hydroxy-pregnenolone and 17alpha-hydroxy-pregnenolone in the foeto-placental unit and mother at midgestation. | Reynolds, JW., et al. 1969. Acta Endocrinol (Copenh). 61: 533-50. PMID: 5820062
- Purification and characterization of three constitutive cytochrome P-450 isoforms from bovine olfactory epithelium. | Longo, V., et al. 1997. Biochem J. 323 (Pt 1): 65-70. PMID: 9173903
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
16α-Hydroxy Progesterone, 25 mg | sc-488185 | 25 mg | $510.00 |
