15(R)-HETE (CAS 83603-31-0)
QUICK LINKS
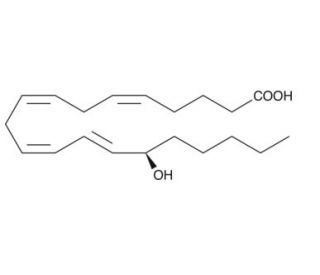
15(R)-HETE, with the CAS number 83603-31-0, is an enantiomer of 15-hydroxyeicosatetraenoic acid, specifically distinguished by the R-configuration of the hydroxyl group at the fifteenth carbon of the eicosanoid chain. This compound is synthesized through the action of 15-lipoxygenase on arachidonic acid. In the realm of biochemical research, 15(R)-HETE is particularly valued for its role in elucidating the mechanisms underlying the regulation of cellular processes such as inflammation, cell signaling, and the modulation of ion channels. The specific actions of 15(R)-HETE include influencing the behavior of various signaling pathways that are critical for cell survival, differentiation, and response to oxidative stress. Researchers use this compound to study its effects on intracellular calcium levels and its interaction with specific receptors and proteins involved in signal transduction. This focus helps to unravel the complex interactions between lipid mediators and cellular mechanisms, enhancing the understanding of how lipid signaling contributes to the regulation of physiological processes. By investigating the functional roles of 15(R)-HETE, scientists gain insights into the detailed molecular interactions that govern cellular responses and adaptations, contributing significantly to the broader field of cellular and molecular biology.
15(R)-HETE (CAS 83603-31-0) References
- 15-Lipoxygenase in glomerular inflammation. | Montero, A. and Badr, KF. 2000. Exp Nephrol. 8: 14-9. PMID: 10644878
- Spatial requirements for 15-(R)-hydroxy-5Z,8Z,11Z, 13E-eicosatetraenoic acid synthesis within the cyclooxygenase active site of murine COX-2. Why acetylated COX-1 does not synthesize 15-(R)-hete. | Rowlinson, SW., et al. 2000. J Biol Chem. 275: 6586-91. PMID: 10692466
- Endogenous anti-inflammatory mediators from arachidonate in human neutrophils. | Vachier, I., et al. 2002. Biochem Biophys Res Commun. 290: 219-24. PMID: 11779156
- Cyclooxygenase-2-mediated DNA damage. | Lee, SH., et al. 2005. J Biol Chem. 280: 28337-46. PMID: 15964853
- Aspirin-triggered 15-epi-lipoxin A4 predicts cyclooxygenase-2 in the lungs of LPS-treated mice but not in the circulation: implications for a clinical test. | Kirkby, NS., et al. 2013. FASEB J. 27: 3938-46. PMID: 23792301
- Aspirin and lipid mediators in the cardiovascular system. | Schrör, K. and Rauch, BH. 2015. Prostaglandins Other Lipid Mediat. 121: 17-23. PMID: 26201059
- Analysis of HETEs in human whole blood by chiral UHPLC-ECAPCI/HRMS. | Mazaleuskaya, LL., et al. 2018. J Lipid Res. 59: 564-575. PMID: 29301865
- Residual cyclooxygenase activity of aspirin-acetylated COX-2 forms 15 R-prostaglandins that inhibit platelet aggregation. | Giménez-Bastida, JA., et al. 2019. FASEB J. 33: 1033-1041. PMID: 30096040
- Characterization of specific subcellular 15-hydroxyeicosatetraenoic acid (15-HETE) binding sites on rat basophilic leukemia cells. | Kang, LT. and Vanderhoek, JY. 1995. Biochim Biophys Acta. 1256: 297-304. PMID: 7786891
- Mutation of serine-516 in human prostaglandin G/H synthase-2 to methionine or aspirin acetylation of this residue stimulates 15-R-HETE synthesis. | Mancini, JA., et al. 1994. FEBS Lett. 342: 33-7. PMID: 8143845
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
15(R)-HETE, 25 µg | sc-205032 | 25 µg | $139.00 | |||
15(R)-HETE, 50 µg | sc-205032A | 50 µg | $199.00 |
