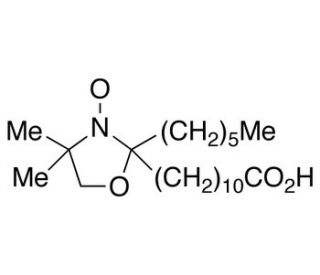
12-Doxylstearic Acid (CAS 29545-47-9)
QUICK LINKS
12-Doxylstearic Acid is a spin label compund, a competitve inhibitor. The stearate spin-labeled at C5 has the highest affinity for the lipoxygenase. The compound known as C10H21NO3, specifically 2-(10-Carboxydecyl)-2-hexyl-4,4-dimethyl-3-oxyloxazolidine, has garnered attention in the fields of biochemistry and physiology due to its potential applications. This innovative synthetic compound has been extensively examined in laboratory applications and research endeavors. C10H21NO3 has demonstrated promise as a stabilizing agent in enzymatic reactions and as a protein degradation inhibitor. Moreover, its interactions with cell membranes and modulation of their properties have been a subject of investigation. Additionally, C10H21NO3 has exhibited antioxidant properties, effectively scavenging free radicals and safeguarding cells against oxidative damage.
12-Doxylstearic Acid (CAS 29545-47-9) References
- Kinetics of inhibition of leukocyte 12-lipoxygenase by the isoform-specific inhibitor 4-(2-oxapentadeca-4-yne)phenylpropanoic acid. | Moody, JS. and Marnett, LJ. 2002. Biochemistry. 41: 10297-303. PMID: 12162745
- Topographical organization of the N-terminal segment of lung pulmonary surfactant protein B (SP-B(1-25)) in phospholipid bilayers. | Wang, Y., et al. 2003. Biochemistry. 42: 4015-27. PMID: 12680754
- Polarity of lipid bilayers. A fluorescence investigation. | Pérochon, E., et al. 1992. Biochemistry. 31: 7672-82. PMID: 1510953
- Beta-blockers and benzodiazepines location in SDS and bile salt micellar systems. An ESR study. | Reis, S., et al. 2007. J Pharm Biomed Anal. 45: 62-69. PMID: 17606356
- Effects of Proteus mirabilis lipopolysaccharides with different O-polysaccharide structures on the plasma membrane of human erythrocytes. | Arabski, M., et al. 2008. Z Naturforsch C J Biosci. 63: 460-8. PMID: 18669036
- Mapping of collision frequencies for stearic acid spin labels by saturation-recovery electron paramagnetic resonance. | Yin, JJ., et al. 1990. Biophys J. 58: 713-20. PMID: 2169919
- Modulation of Na+-Ca2+ exchange and Ca2+ permeability in cardiac sarcolemmal vesicles by doxylstearic acids. | Philipson, KD. and Ward, R. 1987. Biochim Biophys Acta. 897: 152-8. PMID: 3099842
- Lipid peroxidation increases the molecular order of microsomal membranes. | Curtis, MT., et al. 1984. Arch Biochem Biophys. 235: 644-9. PMID: 6097193
- Cholesterol blocks the disordering effects of ethanol in biomembranes. | Chin, JH. and Goldstein, DB. 1984. Lipids. 19: 929-35. PMID: 6527612
- Molecular properties of a stratum corneum model lipid system: large unilamellar vesicles. | Hatfield, RM. and Fung, LW. 1995. Biophys J. 68: 196-207. PMID: 7711242
- Defining the arachidonic acid binding site of human 15-lipoxygenase. Molecular modeling and mutagenesis. | Gan, QF., et al. 1996. J Biol Chem. 271: 25412-8. PMID: 8810309
- (Carboxyalkyl)benzyl propargyl ethers as selective inhibitors of leukocyte-type 12-lipoxygenases. | Gorins, G., et al. 1996. J Med Chem. 39: 4871-8. PMID: 8960545
- Fluorescence studies of exchangeable apolipoprotein-lipid interactions. Superficial association of apolipophorin III with lipoprotein surfaces. | Sahoo, D., et al. 1998. J Biol Chem. 273: 1403-8. PMID: 9430675
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
12-Doxylstearic Acid, 5 mg | sc-206277 | 5 mg | $374.00 |
