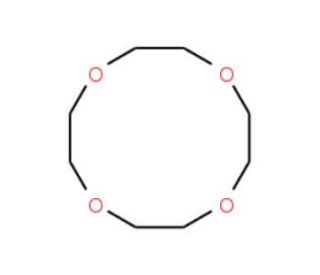
Molecular structure of 12-Crown-4, CAS Number: 294-93-9
12-Crown-4 (CAS 294-93-9)
Application:
12-Crown-4 is an aprotic "solvent" for alkali metals, the sodium solution is a strong reducing agent
CAS Number:
294-93-9
Purity:
≥97%
Molecular Weight:
176.21
Molecular Formula:
C8H16O4
Supplemental Information:
This is as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
12-Crown-4 is a Phase transfer catalyst, complexing-agent for lithium. Aprotic "solvent" for alkali metals, the sodium solution is a strong reducing agent. 12-crown-4, a crown ether, exhibits a unique structure as it is derived from cyclododecane with oxygen atoms replacing the carbon atoms at positions 1, 4, 7, and 10. This intriguing compound belongs to the family of crown ethers and serves as a saturated organic heteromonocyclic parent.
12-Crown-4 (CAS 294-93-9) References
- Formation of molecular radical cations of aliphatic tripeptides from their complexes with CuII(12-crown-4). | Chu, IK., et al. 2004. Rapid Commun Mass Spectrom. 18: 1798-802. PMID: 15329872
- Cationic rare-earth metal trimethylsilylmethyl complexes supported by THF and 12-crown-4 ligands: synthesis and structural characterization. | Elvidge, BR., et al. 2005. Inorg Chem. 44: 6777-88. PMID: 16156637
- Experimental and theoretical study of the vibrational spectra of free 12-crown-4. | El-Azhary, AA. and Al-Kahtani, AA. 2005. J Phys Chem A. 109: 4505-11. PMID: 16833786
- Experimental and theoretical study of the vibrational spectra of 12-crown-4-alkali metal cation complexes. | Al-Rusaese, S., et al. 2006. J Phys Chem A. 110: 8676-87. PMID: 16836428
- Ion-pair extractions with 12-crown-4 and its analogues. | Pacey, GE. and Wu, YP. 1984. Talanta. 31: 165-8. PMID: 18963560
- 7Li NMR chemical shift titration and theoretical DFT calculation studies: solvent and anion effects on second-order complexation of 12-crown-4 and 1-aza-12-crown-4 with lithium cation in several aprotic solvents. | Masiker, MC., et al. 2010. Magn Reson Chem. 48: 94-100. PMID: 19950215
- Complexation of syndiotactic polystyrene with 12-crown-4. | Kaneko, F., et al. 2010. Macromol Rapid Commun. 31: 554-7. PMID: 21590941
- The growth mechanism and ferroelectric domains of diisopropylammonium bromide films synthesized via 12-crown-4 addition at room temperature. | Gao, K., et al. 2016. Phys Chem Chem Phys. 18: 7626-31. PMID: 26956668
- 12-Crown-4 Ether Disrupts the Patient Brain-Derived Amyloid-β-Fibril Trimer: Insight from All-Atom Molecular Dynamics Simulations. | Agrawal, N. and Skelton, AA. 2016. ACS Chem Neurosci. 7: 1433-1441. PMID: 27454141
- Binding of 12-Crown-4 with Alzheimer's Aβ40 and Aβ42 Monomers and Its Effect on Their Conformation: Insight from Molecular Dynamics Simulations. | Agrawal, N. and Skelton, AA. 2018. Mol Pharm. 15: 289-299. PMID: 29200307
- Engineering Li/Na selectivity in 12-Crown-4-functionalized polymer membranes. | Warnock, SJ., et al. 2021. Proc Natl Acad Sci U S A. 118: PMID: 34493651
- Insights of the Structure and Luminescence of Mn2+/Sn2+-Containing Crown-Ether Coordination Compounds. | Bonnin, MA. and Feldmann, C. 2021. Inorg Chem. 60: 14645-14654. PMID: 34520187
- Effect of the Temperature on the Process of Preferential Solvation of 1,4-Dioxane, 12-Crown-4, 15-Crown-5 and 18-Crown-6 Ethers in the Mixture of N-Methylformamide with Water: Composition of the Solvation Shell of the Cyclic Ethers. | Jóźwiak, M., et al. 2023. Int J Mol Sci. 24: PMID: 37240291
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
12-Crown-4, 1 g | sc-254004 | 1 g | $42.00 |
