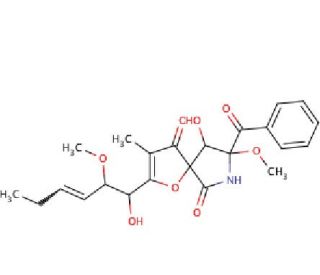
11-O-Methylpseurotin A (CAS 956904-34-0)
QUICK LINKS
11-O-Methylpseurotin A is a secondary metabolite derived from fungi, known for its ability to inhibit chitin synthase, an enzyme critical for the synthesis of chitin in fungal cell walls. The mechanism of action involves binding to the enzyme′s active site, thereby preventing the polymerization of N-acetylglucosamine into chitin, which is essential for maintaining cell wall integrity. This inhibition disrupts the formation of the cell wall, leading to compromised cell structure and, ultimately, cell death in susceptible fungi. In research, 11-O-Methylpseurotin A is utilized to study the biosynthesis and regulation of fungal cell walls. By inhibiting chitin synthase, researchers can investigate the role of chitin in fungal growth, morphogenesis, and pathogenicity. This compound is also used to explore the mechanisms of antifungal resistance, examining how fungi adapt to inhibitors of cell wall synthesis. Additionally, 11-O-Methylpseurotin A serves as a tool in chemical biology for studying the structure-function relationships of chitin synthase and its interactions with other cellular components. These studies contribute to a broader understanding of fungal biology and the development of novel antifungal strategies.
11-O-Methylpseurotin A (CAS 956904-34-0) References
- Pseurotin D Inhibits the Activation of Human Lymphocytes. | Rubanova, D., et al. 2021. Int J Mol Sci. 22: PMID: 33669259
- Discovery of novel secondary metabolites encoded in actinomycete genomes through coculture. | Kim, JH., et al. 2021. J Ind Microbiol Biotechnol. 48: PMID: 33825906
- In silico molecular docking of SARS-CoV-2 surface proteins with microbial non-ribosomal peptides: identification of potential drugs. | Bansal, P., et al. 2021. J Proteins Proteom. 12: 177-184. PMID: 34456530
- Quantitative Morphological Analysis of Filamentous Microorganisms in Cocultures and Monocultures: Aspergillus terreus and Streptomyces rimosus Warfare in Bioreactors. | Ścigaczewska, A., et al. 2021. Biomolecules. 11: PMID: 34827738
- Impact of Co-Culture on the Metabolism of Marine Microorganisms. | Caudal, F., et al. 2022. Mar Drugs. 20: PMID: 35200682
- Discovery of Anti-MRSA Secondary Metabolites from a Marine-Derived Fungus Aspergillus fumigatus. | Zhang, R., et al. 2022. Mar Drugs. 20: PMID: 35621953
- Anti-inflammatory alkaloids from the cold-seep-derived fungus Talaromyces helicus SCSIO41311. | Cong, M., et al. 2022. 3 Biotech. 12: 161. PMID: 35818470
- Biomolecules in modern and sustainable agriculture. | Schmaltz, S., et al. 2023. 3 Biotech. 13: 70. PMID: 36742447
- Elicitation for activation of the actinomycete genome's cryptic secondary metabolite gene clusters. | El-Hawary, SS., et al. 2023. RSC Adv. 13: 5778-5795. PMID: 36816076
- Bioactive Alkaloids as Secondary Metabolites from Plant Endophytic Aspergillus Genus. | Zhu, J., et al. 2023. Molecules. 28: PMID: 38067519
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
11-O-Methylpseurotin A, 250 µg | sc-391080 | 250 µg | $60.00 |
