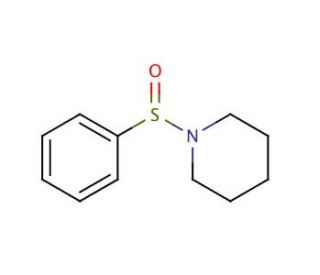
1-(Phenylsulfinyl)piperidine (CAS 4972-31-0)
QUICK LINKS
1-(Phenylsulfinyl)piperidine is a piperidine compound with an organic structure that includes a sulfur atom and a nitrogen atom within its five-membered ring. This versatile compound has found extensive utility in scientific research, serving as a ligand in organometallic complexes, an enzyme inhibitor, and a fundamental building block for synthesizing various compounds. Its biological significance extends to biochemical and physiological effects in living organisms. Notably, it acts as an inhibitor of enzymes like acetylcholinesterase, which participates in acetylcholine breakdown. Furthermore, 1-(Phenylsulfinyl)piperidine has proven invaluable as a precursor for synthesizing compounds like 1-phenylsulfinyl-2-piperidinone.
1-(Phenylsulfinyl)piperidine (CAS 4972-31-0) References
- Synthesis of n-octyl 2,6-dideoxy-alpha-L-lyxo-hexopyranosyl-(1-->2)-3-amino-3-deoxy-beta-D-galactopyranoside, an analog of the H-disaccharide antigen. | Bai, Y., et al. 2006. Carbohydr Res. 341: 1702-7. PMID: 16616902
- Programmable reactivity-based one-pot oligosaccharide synthesis. | Lee, JC., et al. 2006. Nat Protoc. 1: 3143-52. PMID: 17406577
- Strong aphicidal activity of GlcNAc(β1→4)Glc disaccharides: synthesis, physiological effects, and chitinase inhibition. | Dussouy, C., et al. 2012. Chemistry. 18: 10021-8. PMID: 22764072
- Studies related to Norway spruce galactoglucomannans: chemical synthesis, conformation analysis, NMR spectroscopic characterization, and molecular recognition of model compounds. | Ekholm, FS., et al. 2012. Chemistry. 18: 14392-405. PMID: 23008171
- Thin-layer chromatography enantioseparations on chiral stationary phases: a review. | Del Bubba, M., et al. 2013. Anal Bioanal Chem. 405: 533-54. PMID: 23161065
- A New, Practical One-Pot Synthesis of Unprotected Sulfonimidamides by Transfer of Electrophilic NH to Sulfinamides. | Izzo, F., et al. 2017. Chemistry. 23: 15189-15193. PMID: 28833686
- Comparison of disaccharide donors for heparan sulfate synthesis: uronic acids vs. their pyranose equivalents. | Sheppard, DJ., et al. 2020. Org Biomol Chem. 18: 4728-4733. PMID: 32531013
- 2,3-Carbamate mannosamine glycosyl donors in glycosylation reactions of diacetone-d-glucose. An experimental and theoretical study. | Morelli, L., et al. 2021. Carbohydr Res. 509: 108421. PMID: 34450528
- Accessing Perfluoroaryl Sulfonimidamides and Sulfoximines via Photogenerated Perfluoroaryl Nitrenes: Synthesis and Application as a Chiral Auxiliary. | Proietti, G., et al. 2021. J Org Chem. 86: 17119-17128. PMID: 34766772
- Convergent synthesis of oligomannose-type glycans via step-economical construction of branch structures. | Sano, K., et al. 2023. Carbohydr Res. 525: 108764. PMID: 36812846
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1-(Phenylsulfinyl)piperidine, 5 g | sc-222629 | 5 g | $230.00 |
